Translate this page into:
A successful story of translational orthodontic research: Micro-osteoperforation-from experiments to clinical practice
Address for Correspondence: Dr. Michelle Yuching Chou, Department of Developmental Biology, Harvard School of Dental Medicine, Boston, Massachusetts, USA. E-mail: ychou@post.harvard.edu
This article was originally published by Wolters Kluwer and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The gap between basic science research and clinical application has long existed and therefore translational research has emerged in recent years to bridge such gap. Consortium for Translational Orthodontic Research (CTOR) was established with missions to integrate resources from different entities and to provide a platform for interdisciplinary groups who share the same vision to exchange ideas and inspire innovations. During its short existence, CTOR has successfully carried out several research projects which led to various innovations. Micro-osteoperforation is by far one of the most successful examples of translational research in the orthodontic field. It exemplifies how translational research can benefit scientists, clinicians, and patients. In this article, the process of its development, the rationale and scientific evidence from animal and clinical studies, and how it can be applied in daily practice will be depicted.
Keywords
Accelerated orthodontics
accelerated tooth movement
biology of tooth movement
Consortium for Translational Orthodontic Research (CTOR)
micro-osteoperforation (MOP)
translational research
INTRODUCTION
A long-standing pitfall in medical and dental research is the big gap between basic science research and clinical practice. Therefore, in recent years, translational research has emerged to bridge between the two. Translational research is a highly interdisciplinary field that integrates disciplines, resources, expertise, and techniques within these pillars to promote enhancements in prevention, diagnosis, and therapies [Figure 1].[1]
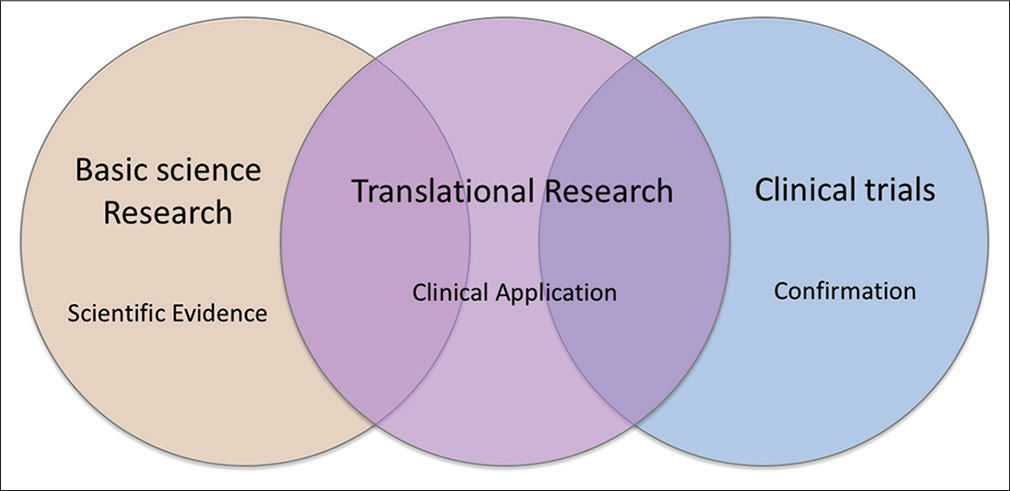
- Translational research integrates resources in different fields and bridges the gap between basic science and clinical practice
The sense of urgency of bridging between basic science research and clinical application in orthodontics has motivated us to launch the only center in the world dedicated to translational orthodontic research, the Consortium for Translational Orthodontic Research (CTOR; https://orthodonticscientist.org). We believe that by establishing a series of structurally designed bench, animal, and clinical studies, dental scientists eventually can make meaningful use of the laboratory research, and translate science into the advancement of clinical care. Ultimately, both patients and health-care providers can benefit from the improvement of clinical care.
Since its establishment in 2009, CTOR has integrated the resources and input from collaborators in basic science, clinical practice and industry. It also has provided a platform for interdisciplinary groups who share the same vision of improving the quality of care to exchange ideas and inspire innovations [Figure 2]. To date, the translational research conducted by CTOR have led to 7 United States patents, with the main focus of developing new modalities for safer and faster orthodontic treatment, expanding the boundaries of orthodontic correction and orthopedic treatment, and growing/retaining bone in the jaws. These patents have attracted companies to invest in further research and development (R&D), and also commercializing CTOR’s intellectual properties.
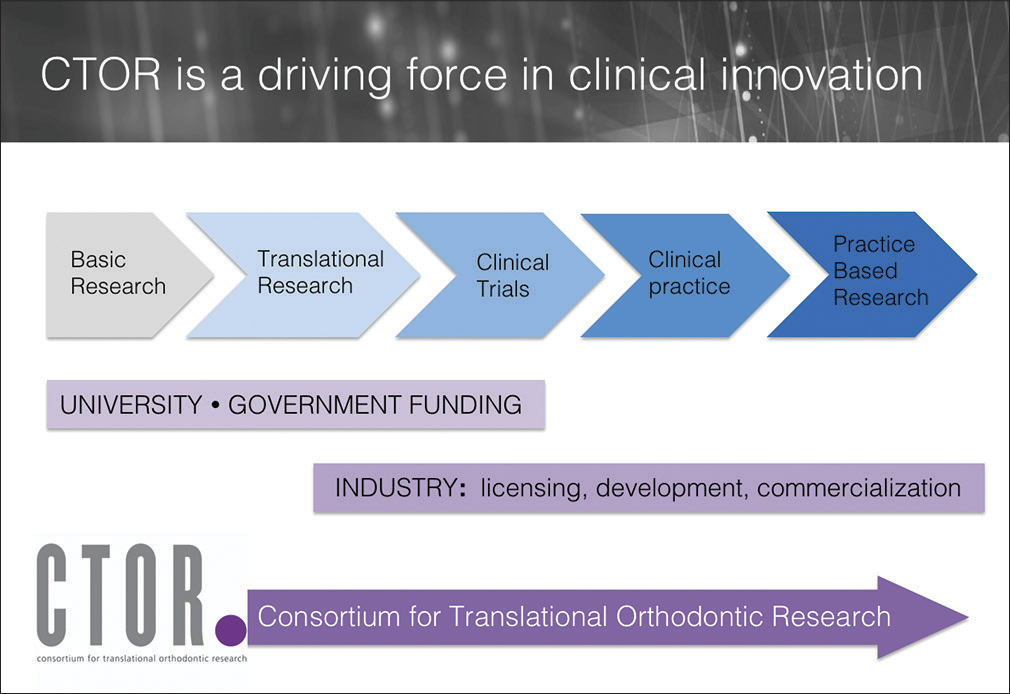
- The mission of Consortium for Translational Orthodontic Research is to integrate basic science, clinical science, and industrial resources in the field of orthodontics. It functions as a driving force in advancement of orthodontics
One of the most successful productions by CTOR is the innovative concept and device, micro-osteoperforation (MOP), which was originally designed to facilitate orthodontic treatment. It has been commercialized and is gaining popularity in the global market for its efficiency and effectiveness in accelerating orthodontic treatment, and its versatility in clinical applications.
In this article, we would like to share the story of how MOP technique was successfully developed, from conceptualization, bench study, clinical study, to now available for orthodontic clinicians on all continents. We will summarize the scientific evidence in the biological mechanism, efficiency, and effectiveness of MOP. The basic guidelines on how to apply this technique in daily practice will also be reviewed.
HISTORICAL BACKGROUND
MOP is a concept and technique developed by CTOR less than a decade ago. Its effectiveness and efficiency in accelerating tooth movement were first proven by animal and human studies conducted by scientists and clinicians at CTOR. The technique/device was then patented by CTOR, and licensed to Propel Orthodontics (Ossining, NY, USA) to commercialize the device. Since Propel’s establishment in 2010, they have had ever-increasing sales in the global market, in a scope of multimillion US dollars annually.
RATIONALE AND DEVELOPMENT OF MICRO-OSTEOPERFORATION CONCEPT
Over the past decade, accelerated orthodontics has become one of the most extensively researched and marketed areas in the orthodontic arena, and it still has much potential to grow. Several techniques have claimed to improve treatment efficiency, that is, especially beneficial for shortening treatment duration in complex adult treatment. As a result, these techniques have been marketed as practice building tools. However, not all of these techniques have been proven effective scientifically.
On the other hand, as a matter of fact, the level of scientific evidence that MOP bears is one of the highest among all accelerated techniques. Therefore, it is able to provide clinicians promising and consistent clinical outcomes. In the following sections, we will demonstrate the conceptualization and progression of scientific evidence of MOP.
Biology of orthodontic tooth movement
Biological response plays a central role in controlling orthodontic tooth movement.[2] Force application on teeth induces an aseptic, acute inflammatory response that involves a series of molecular and cellular events, resulting in osteoclast formation and activation that initiates bone remodeling machinery and allows movement of teeth to occur. During the inflammatory response, a repertoire of chemokines and cytokines are activated thus inflammatory cells and osteoclast precursors are recruited into the area; in turn, more inflammatory markers are released which directly or indirectly activate RANK-RANKL pathway toward osteoclast differentiation and activation [Figure 3]. Therefore, the rate of tooth movement is determined by the rate of bone resorption in the direction of movement; meanwhile, the rate of bone resorption is determined by the rate of osteoclast formation. The positive correlation between the activities of inflammatory marker, osteoclast activation, and rate of tooth movement has been demonstrated in our previous study in rats and humans.[3-5]
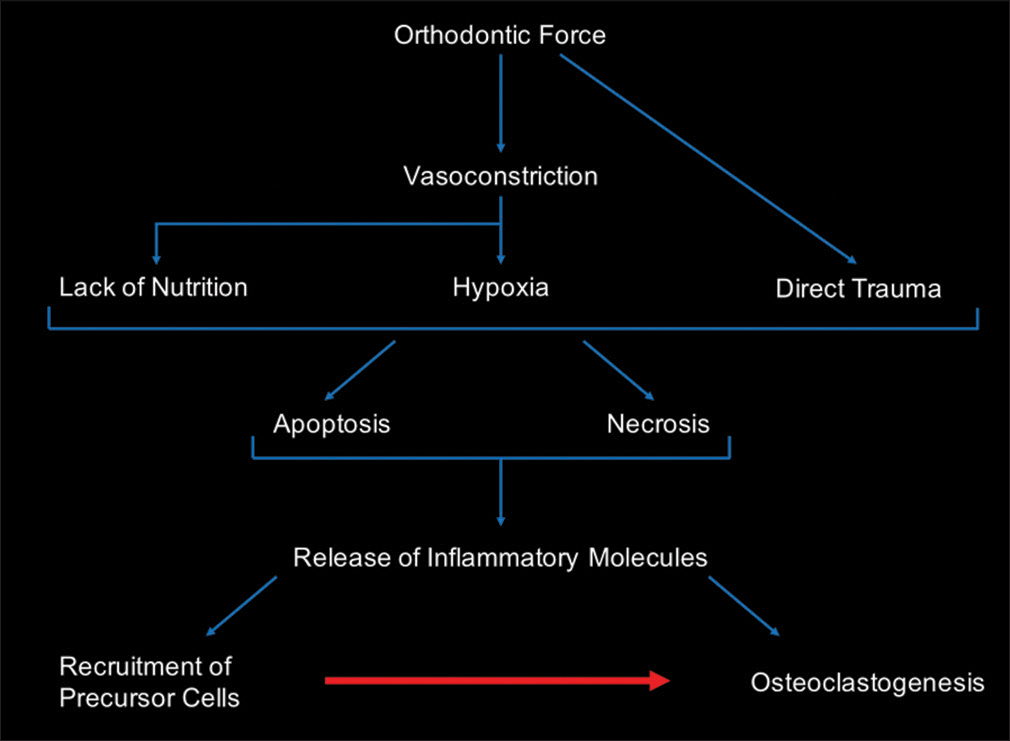
- Diagram of cellular events in the compression side in response to application of orthodontic force
The importance of cytokines in controlling the rate of tooth movement can be appreciated from studies that block their effects. It has been shown that injection of interleukin-1 receptor antagonist or soluble tumor necrosis factor (TNF)-α receptor type I antagonist results in a 50% reduction in tooth velocity.[6-8] Similarly, tooth movement in TNF receptor type II-deficient mice is reduced compared to wild-type mice.[9] Animals deficient in CC chemokine receptor 2 (i.e., the receptor for chemokine ligand 2) or chemokine ligand 3 show a significant reduction in orthodontic tooth movement and the number of osteoclasts.[10] Likewise, it is well known that nonsteroidal anti-inflammatory drugs can reduce the velocity of tooth movement by inhibiting prostaglandin synthesis.[11,12] Inhibition of other derivatives of arachidonic acid, such as leukotrienes, also significantly decreases the rate of tooth movement.[13]
These studies strongly support that the proinflammatory cytokines are essential mediators of orthodontic tooth movement. Such findings have compelled us to develop a simple method to amplify and titrate the patient’s normal biological response to safely accelerate orthodontic tooth movement.
Boosting the biological response to orthodontic force
Based on the understanding of normal biological events on the application of orthodontic force, it is logical to assume that increasing the amount of inflammatory mediators should increase the rate of tooth movement. Therefore, developing an approach to stimulate the body to produce an increased amount of inflammatory mediator is the fundamental drive during the R&D process of accelerated techniques.
One of the most intuitive and common measures among clinicians to conquer slow tooth movement is to increase the magnitude of orthodontic force. Our studies at CTOR demonstrated that increasing the magnitude of orthodontic force increases inflammatory marker levels, osteoclast recruitment and formation, alveolar bone resorption, and the rate of tooth movement. However, there is a force level above which we cannot stimulate these biological responses any further.[3] Thus, the magnitude of cytokine release that can be induced by orthodontic forces has an upper limit and consequently the osteoclast activity initiated by orthodontic forces has a “biological saturation point.” While increasing the force magnitude does not overcome this limitation, it requires another methodology that can increase the osteoclast numbers in the area to enhance this biological response.
Some studies have attempted to inject prostaglandins[14] or arachidonic acid derivatives[15] locally or systemic application of PGE1 analog[16] to increase the rate of tooth movement. Although such measures do increase the rate of tooth movement, the short half-life and side effects such as hyperalgesia have limited its clinical application.
Another approach to increase the proinflammatory mediators is to stimulate the body to produce these factors at a higher level through its natural course. The advantage of such approach is a coordinated increase in the level of all inflammatory mediators. As discussed before, many cytokines participate in response to orthodontic forces. Injecting one cytokine does not mimic the normal inflammatory response, which is a balance of pro- and anti-inflammatory mediators. Imbalance of such reactions can lead to many side effects, thus it is unsafe to be utilized in daily clinical practice.
Micro-osteoperforation – Evidence from rats to humans
The concept of MOP originated from the attempt of boosting normal inflammatory responses, during which pro-inflammatory mediators are stimulated then followed by an appropriate recovery/repair process. Our animal study that was first published in Journal of Dental Research has shown that introducing small perforations in the alveolar bone (micro-osteoperforation) during orthodontic tooth movement can significantly stimulate the expression of inflammatory mediators. While the application of orthodontic force beyond the saturation point does not elevate the expression and activation of inflammatory mediators beyond certain levels,[3] adding MOP to the area of tooth movement increases the level of inflammatory mediators.[5] This response is accompanied by a significant increase in the osteoclast number, bone resorption and localized osteopenia around all adjacent teeth, which could explain the increase in the rate of tooth movement.
Similarly, our human clinical trial using a canine retraction model demonstrated that MOP can amplify the catabolic response to orthodontic forces. Canine retraction in the presence of MOP results in twice as much distalization compared with patients receiving similar orthodontic forces without MOP. This acceleration in tooth movement is accompanied by an increase in the level of inflammatory mediators.[17]
In addition, our clinical studies demonstrated that increasing the number of MOP significantly increases expression of inflammatory mediators and the magnitude of tooth movement.[18] Therefore, one should expect procedures such as orthognathic surgery, corticotomies, or piezocision to significantly increase the levels of inflammatory cytokines beyond those induced by MOP. Although increase in cytokine release by these methods is accompanied with a higher rate of tooth movement, the increase in the expression of inflammatory mediators is not sustained for a long time, unfortunately. A significant decrease in cytokine activity is observed 2–3 months after any of these treatments. As a result, each of these procedures would need to be repeated during orthodontic treatment, which renders some of the above-mentioned modalities impractical.
CLINICAL APPLICATIONS OF MICRO-OSTEOPERFORATION
Orthodontic mechanotherapy is made up of carefully designed stages that lead to an optimal treatment outcome. At certain stages, some teeth are targeted to move while others are designed to serve as anchors. MOP can easily be incorporated into our orthodontic mechanics. It can be selectively applied to target areas to enhance tooth movement in one region while preventing anchorage loss in another as treatment dictates.
The progressive nature of orthodontic treatment mechanics renders procedures that can only be applied once or twice throughout the treatment, such as corticotomy and piezocision, unfavorable. These procedures can cause more extensive trauma in broader areas, therefore stimulating a higher amount of proinflammatory cytokines. Although they can be preferable in certain clinical situations, the level of inflammatory markers decreases significantly 2–3 months after surgery. Therefore, if a longer distance of tooth movement is required, MOP is the procedure of choice as it can be applied periodically until the desired movement is achieved.
Another clinical advantage of MOP is the ability to titrate the rate by establishing “Biological Anchorage” while accelerating the movement of target teeth. Application of MOP on particular locations can selectively decrease the bone density around the target tooth while the bone density around the anchor unit remains unchanged. This results in titrated rates of tooth movement among two different units.
MOP can also facilitate root movement, which is deemed to be the most difficult movement to accomplish in orthodontics. By activating osteoclasts and decreasing the bone density, MOP can decrease the stress on the root during movement and therefore decrease the possibility of root resorption. Applying the same logic, MOP should be considered during segmental intrusion in adults, during which there is a possibility of root resorption as a result of the high-stress root apices bear.
More details in clinical application of MOP can be found in our newest book “Clinical Guide to Accelerated Orthodontics.”[19] More details in fundamental and advanced mechanotherapy can be found in “Mechanotherapy in Orthodontics.”[20] It should be emphasized that regardless of which accelerated technique is chosen, sound biomechanical plans should never be overlooked.
FUTURE OUTLOOK OF MICRO-OSTEOPERFORATION
Although the application of MOP in accelerated orthodontics is gaining popularity, its usage is not limited to accelerating tooth movement. As scientific evidence expands and more translational research is conducted at CTOR, we discovered a variety of clinical applications that can generate revolutionary changes in our specialty. In our most recently published book chapter, we introduced a new theory on the biology of tooth movement – “Biphasic Theory.”[21] We revealed the scientific evidence on biological events of tooth movement in two phases, the catabolic and anabolic phase. These two phases have a specific chronological order. This in turn has proven that certain statements in the existing literature are misleading. More importantly, we discovered that based on Biphasic Theory, MOP can indeed expand treatment boundaries in orthodontics. It allows us to treat adult, complex cases which were deemed surgical cases with a nonsurgical treatment option.
Application of micro-osteoperforation in edentulous ridge and extensive sinus pneumatization
A simplified description of biphasic theory is that activation of osteoblasts by osteoclasts is observed during tooth movement where the bone resorption phase of tooth movement (catabolic phase) is followed by a bone formation phase (anabolic phase) to prevent bone loss during tooth movement. A similar phenomenon can be stimulated during movement of a tooth into an area of alveolar bone loss. These areas usually are occupied with a thick cortical bone that is short in height and narrow in width. Moving a tooth in this area is usually extremely slow, can cause root resorption and usually results in tilting the crown into the edentulous space without significant root movement. Applying MOP in this edentulous area harnesses the catabolic phase of orthodontic treatment to decrease the bone density. This allows faster tooth movement into the area with less possibility of root resorption and greater bodily movement rather than tipping. This osteoclast activity then increases osteoblast activity significantly, which couples catabolism-dependent tooth movement with anabolism-dependent remodeling that restores the bone height and width in the previously edentulous site. Such phenomenon can be utilized for implant site development or tooth movement into maxillary edentulous areas that have extensive sinus pneumatization.[19]
Micro-osteoperforation-generated cortical drift
Alveolar cortical bone sets the physical and physiological limits of orthodontic tooth movement. While a tooth can be driven through the cortical plate if the orthodontic force applied to it has sufficient magnitude, direction and duration, the speed of cortical bone remodeling is slow enough that appropriately directed forces rarely place any tooth in danger of breaching the physical limit set by the cortical bone. However, orthodontists face a conundrum when they have a borderline extraction case where expansion would provide the ideal space needed to unravel the crowding, but the alveolar boundary conditions are not robust enough to tolerate the expansion. Therefore, it would be beneficial for orthodontists to manipulate these boundary conditions by increasing bone formation at the surface of the cortical bone. Application of MOP in the opposite direction of orthodontic tooth movement can stimulate osteoclasts that will first decrease the bone density of cortical bone, and second stimulate osteoblast activity in the direction of movement. This treatment results in the drifting of the cortical plate into a new position with significant bone formation in the direction of tooth movement. This is especially important during movement of teeth toward the cortical boundaries, for example, during expansion in adults or retraction of lower anterior teeth during correction of severe class III patients.[19]
TRANSFORMATION TO BE CONTINUED
The mission of CTOR is to build a bridge between academia and industry to allow both parties to advance and eventually improve clinical care. It offers an open environment within which basic scientists and clinicians can interact, exchange ideas, select and pursue research in specific areas of Craniofacial Biology and Orthodontics. MOP is merely one of the successful models of CTOR’s innovations. There are many other areas in orthodontics that are worth exploring. CTOR has been conducting various studies including in vitro studies, animal studies and clinical trials to advance our understanding in key areas of orthodontic and bone biology research, such as molecular orthodontics, mechanotransduction, accelerated tooth movement, craniofacial development, tissue engineering, regulation of skeletogenesis, and gene therapy. The results of these studies have been extensively published in major peer-reviewed journals and books.
In addition, as technology advances and new techniques emerge, every practitioner will need periodically updated training at some point of their careers. Therefore, CTOR not only publishes education material[19,20] but provides continuing education courses and fellowship opportunities for practitioners, residents, and faculty around the world who seek basic and/or advanced training in research and clinical orthodontics.
SUMMARY
In this article, we demonstrated how CTOR developed an innovative technique and device by “translating” basic science to clinical application. We selected MOP as our model to demonstrate how CTOR function as a driving force in this innovation, from conceptualization, conducting basic research and clinical trials, and eventually commercializing the device. This has expedited the realization process of MOP and therefore clinicians and patients around the world can benefit from scientific research. In addition, we believe that as technology advances and new techniques emerge, continuous learning to keep oneself up to date is essential for every clinician to provide the best care to all the patients.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Translational medicine definition by the european society for translational medicine. New Horiz Transl Med. 2015;2:86-8.
- [CrossRef] [Google Scholar]
- Biological Mechanisms to Accelerate Tooth Movement In: Ramalingam AVSS, ed. Stem Cell Biology and Tissue Engineering in Dental Sciences. Boston: Academic Press; 2015. p. :787-98. Ch. 58 Available from: http://www.sciencedirect.com/science/article/pii/B978012397157900062X [Last accessed on 2017 Jan 11]
- [Google Scholar]
- Saturation of the biological response to orthodontic forces and its effect on the rate of tooth movement. Orthod Craniofac Res. 2015;18(1):8-17.
- [Google Scholar]
- Effect of micro-osteoperforations on the rate of tooth movement. Am J Orthod Dentofacial Orthop. 2013;144:639-48.
- [Google Scholar]
- Human interleukin-1 beta and interleukin-1 receptor antagonist secretion and velocity of tooth movement. Arch Oral Biol. 2001;46:185-9.
- [CrossRef] [Google Scholar]
- The role of tumor necrosis factor receptor type 1 in orthodontic tooth movement. J Dent Res. 2007;86:1089-94.
- [CrossRef] [PubMed] [Google Scholar]
- Soluble cytokine receptor treatment in experimental orthodontic tooth movement in the rat. Eur J Orthod. 2005;27:1-11.
- [Google Scholar]
- Experimental model of tooth movement by orthodontic force in mice and its application to tumor necrosis factor receptor-deficient mice. J Bone Miner Metab. 2006;24:20-7.
- [CrossRef] [PubMed] [Google Scholar]
- Role of CCR2 in orthodontic tooth movement. Am J Orthod Dentofacial Orthop. 2012;141:153-60.
- [Google Scholar]
- Non-steroidal and steroidal anti-inflammatory use in the context of orthodontic movement. Eur J Orthod. 2012;34:531-5.
- [CrossRef] [PubMed] [Google Scholar]
- The effect of indomethacin (an aspirin-like drug) on the rate of orthodontic tooth movement. Am J Orthod. 1986;89:312-4.
- [Google Scholar]
- Leukotrienes in orthodontic tooth movement. Am J Orthod Dentofacial Orthop. 1989;95:231-7.
- [CrossRef] [Google Scholar]
- Comparison of the effects of 1,25 dihydroxycholecalciferol and prostaglandin E2 on orthodontic tooth movement. Am J Orthod Dentofacial Orthop. 2004;125:607-14.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of PGI2 and TxA2 analogs and inhibitors in orthodontic tooth movement. Angle Orthod. 2004;74:526-32.
- [Google Scholar]
- Effect of misoprostol, a prostaglandin E1 analog, on orthodontic tooth movement in rats. Am J Orthod Dentofacial Orthop. 2002;122:542-7.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of micro-osteoperforations. Authors’ response. Am J Orthod Dentofacial Orthop. 2014;145:273-4.
- [Google Scholar]
- Micro-osteoperforations: Minimally invasive accelerated tooth movement. Seminars in Orthodontics. 2015;21:162-9.
- [CrossRef] [Google Scholar]
- Clinical Guide to Accelerated Orthodontics: With a Focus on Micro-Perforation. Cham: Springer International Publishing AG; 2017.
- Mechanoterapy in Orthodontics. Hoboken: CTOR Press; 2015.
- Biphasic theory of tooth movement: Cytokine expression and rate of tooth movement In: Shroff B, ed. Biology of Orthodontic Tooth Movement: Current Concepts and Applications in Orthodontic Practice. Cham: Springer International Publishing; 2016. p. :45-65.
- [Google Scholar]






