Translate this page into:
Custom fabrication of cast splint microimplant-assisted rapid palatal expansion appliances for treatment of maxillary deficiency in young adults

*Corresponding author: Hui Xiong, Department of Orthodontics, Hubei-MOST KLOS & KLOBM, School and Hospital of Stomatology, Wuhan University, Wuhan, Hubei, China. xiongh76@163.com
-
Received: ,
Accepted: ,
How to cite this article: Almaqrami BS, Saireke H, Xiying W, Ngan P, Hui C, Xiong H, et al. Custom fabrication of cast splint microimplant-assisted rapid palatal expansion appliances for treatment of maxillary deficiency in young adults. APOS Trends Orthod 2021;11:247-53.
Abstract
Background:
The objective of this article was to illustrate the process in the fabrication of a custom cast splint Microimplant-Assisted Rapid Palatal Expansion (MARPE) appliance for treatment of maxillary deficiency in young adults.
Findings:
The fabrication of a custom-made MARPE appliance allows the casting base to design according to the shape of the patient’s palate close to the palatal mucosa. In this new design, one end consists of a casting body, inserting hole, and teeth retention device (circumferential rings) connected to the other end by a jackscrew. Connection of all parts in the integral casting unit increases the strength of the expansion appliance and avoids distortion, deformation, or damage to the palatal mucosa due to increased resistance from the oromaxillofacial area.
Conclusion:
This custom design can improve the clinical outcome of maxillary expansion, especially in patients with narrow and high-arched palates.
Keywords
Transverse deficiency
Microimplant-assisted rapid palatal expansion design
Maxillary expansion in adolescent patients
INTRODUCTION
Adolescent and adult patients with transverse skeletal deficiencies are at an increased risk of developing temporomandibular joints, musculature, periodontal tissue, and airway disorders, showing more signs and symptoms of these problems in the susceptible patient.[1-3] The exaggerated Curve of Wilson resulting from maxillary transverse deficiency may play a role in centric relation/central occlusion discrepancies, adverse periodontal stresses, and craniofacial development.[3] Therefore, normalizing the transverse jaw relationship in young patients is essential for stable, well-balanced, and proper functional occlusion.
Different appliance designs and techniques have been advocated in the literature to enhance the skeletal contribution to palatal expansion in growing patients. In non-growing young adults, several authors have suggested using microimplants as skeletal anchorage to optimize the application of mechanical forces to circummaxillary sutures, thus avoiding the need for surgical osteotomies.[4] There are various designs of the Microimplant-Assisted Rapid Palatal Expansion (MARPE) appliance. The Maxillary Skeletal Expander (MSE) was designed and proposed by Moon is a “hybrid” tooth-borne and bone-borne appliance with the jet screw connected to the permanent first molars.[5-7] The Bone Acrylic Maxillary Expander appliance is a pure bone-borne appliance with force applied only to the microimplants, and not anchor to any teeth or periodontium.[5]
The insertion slots of the MSE and several other MARPE appliances are located at the four corners of the jet screw.[6-8] Since the chance of microimplant deformation is higher if the force is applied farther from the implant/bone interface and the placement of the jackscrew is almost fixed to the posterior hard palate, it would be nice if the appliance can be custom made. In addition, there is a transverse suture between maxillary palatine processes and the horizontal osseous laminae of the palatal bone, and the miniscrews secured too posteriorly may be located too close to these structures. Furthermore, in continuity with the soft palate, the posterior hard palate has several small salivary glands. Miniscrews that were placed too posteriorly might affect such glands, thus provoking a mucus-retention phenomena similar to what occurs with oral mucocele and/or necrotizing sialometaplasia.[6] At the posterior lateral portion of the hard palate, near the posterior alveolar process, there are also nerves and vessels which emerge from the palatine foramina. One additional limitation of commercially available MSE appliances is placement in patients with narrow and high-arched palates. The proper vertical positioning and bicortical bone penetration may be restricted and could reduce the success rate. In these designs, tipping off the microimplants can still occur to a certain extent because of the small gap between the microimplant and the anterior surfaces of the insertion slots.
The objective of this paper was to describe a custom-made cast splint MARPE appliance that may overcome a few of these shortcomings. The custom-made maxillary skeletal expander includes a jackscrew, four inserting holes, a casting base, and teeth retention device. The two parts of inserting holes, casting base, and the teeth retention device connect together by jackscrew expander that is welded and connected with the casting body.
MATERIAL AND METHODS
Laboratory and clinical procedures
The laboratory fabrication of the custom-made MARPE appliance commenced with a thorough explanation of treatment procedures to the patients, clarifying all details, and technical limitations. This was followed by taking a conventional alginate transfer impression and regular stone plaster pouring.
A cone-beam computed tomography (CBCT) was used to determine the thickness of the hard tissue palate and to identify a suitable implant placement site. A periodontal probe was used to measure the thickness of the soft tissue to determine the appropriate implant length (8, 10, 14 mm, A1, SYNTEC SCIENTIFIC CORPORATION, Taiwan, China) taking into account the thickness of the hard and soft tissue in addition to 2 mm height of the inserting holes.
The size of the jack screw to be used for expansion (8-, 10- or 12-mm, Snap-Lock-Expander, Forestadent, Germany) was selected according to the required amount of expansion. After determining the suitable implantation sites on the CBCT scan, it was drawn on the cast model and waxed up together with the pre-determined position of the four inserting holes (diameter 2 mm, and height 2 mm). The cast maxillary skeletal expander with the base, inserting holes, and the teeth retention part were then welded to the jack screw before finishing and polishing [Figure 1]. The clinical procedure consisted of cementation of the appliance in patient, checking the vertical position of the appliance in relation to the palate. Topical and local anesthesia were administered and microimplants were placed using the self-drilling method with appropriate digital key (Syntec scientific Taiwan, China). The microimplants were placed carefully even though the direction of insertion was guided by the vertical length of the microimplant slots. Microimplants were placed as perpendicular as possible to the palatal bone as well as to each other for effective force distribution. Both the anteroposterior and the lateral inclination were repeatedly checked during placement. Ligature wire was used to tie the microimplant of the same side together to prevent any microimplant from loosening and fell off in the mouth. Expansion activation with a rate of 2–3 turns/day (1/4mm/ turn) was given to the patient together with oral hygiene instructions. Prescription of analgesic drugs and antibiotics was given as needed depending on the patient’s general health.
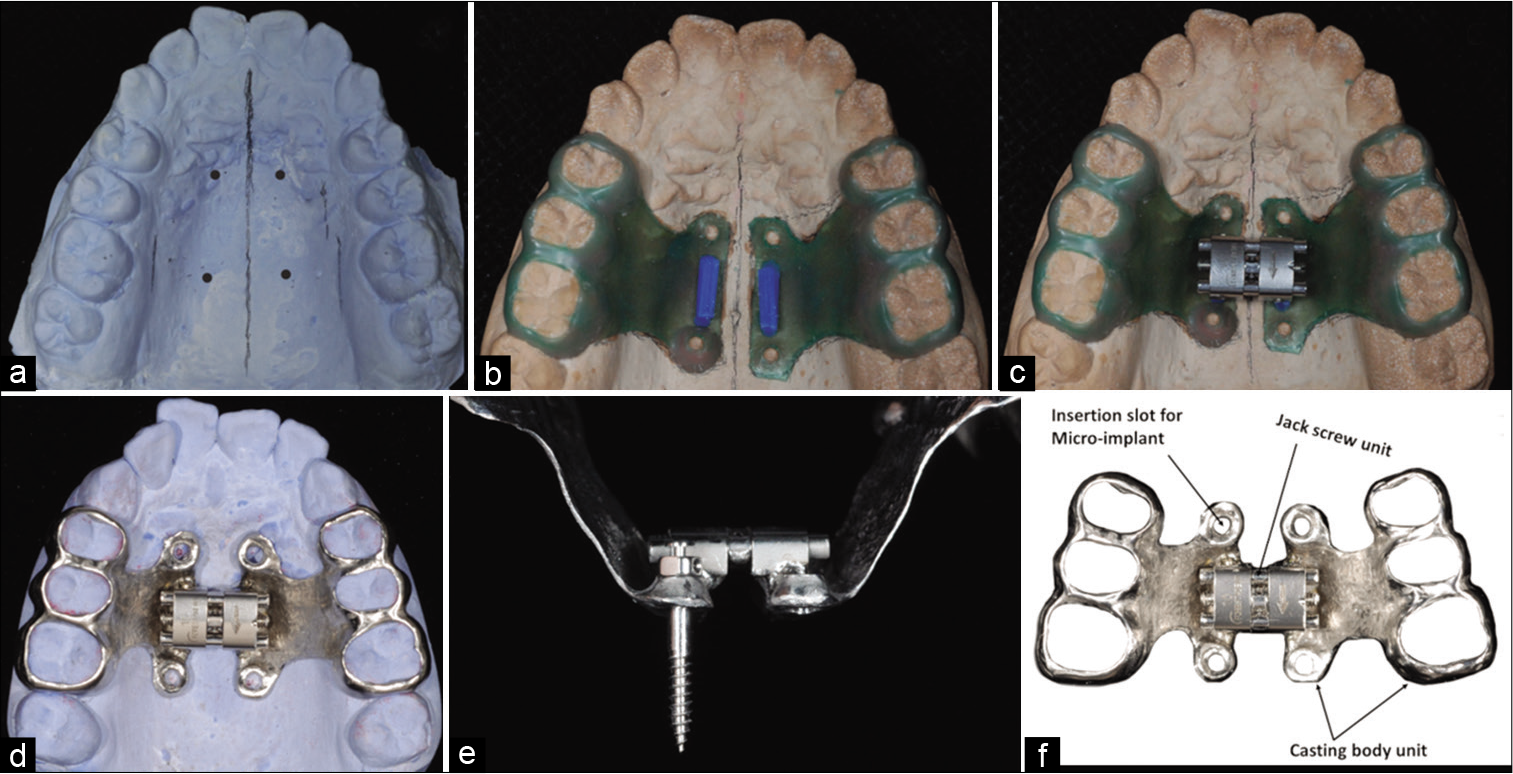
- Laboratory procedures in fabricating a custom-made maxillary skeletal expander. (a) using cone-beam computed tomography to determine the implantation sites of the four micro-implants, (b) wax-up of the base with the correct position of the four inserting holes and connected to the teeth retention part, (c) connecting the jackscrew to the base as one unit before casting, (d) finishing of the custom-made cast splint expander, (e) placement of micro-implants in the casted slots, (f) different parts of the cast splint expander.
Patients were instructed to return for a follow-up visit at least once a week. If the patient cannot activate the expander at home due to increased resistance, activation will be done in office by the clinician. Expansion protocol was carried out according to the skeletal maturation of the patient, and the stability of microimplants were checked every visit. If the microimplant became loose, they will be removed. After maxillary expansion, flowable composite resin was used to fix the screw for 6–9 months.
Removal of microimplants was performed with the same connector coupled with the digital key and turned slowly in a counterclockwise direction. Plaque accumulation on the microimplant head might hinder the grip of the microimplants. In most instances, the microimplants should be able to remove without local anesthesia. After removal of the microimplants, a cotton pellet soaked in hydrogen peroxide was applied to the site to promote asepsis, but no additional care was required. Mucosa wounds usually healed in 2–3 days. Microimplants should be discarded after use and should never be sterilized or reused.
In a custom-made maxillary skeletal expander, the casting base is designed according to the shape of the patient’s palate, close to the palatal mucosa. This design will effectively be preventing the microimplants from bending, loosening or falling off. In addition, with reduced costs, ease of access to lab support, turnover time and far fewer risks than alternative treatment options, offer great promise for the future of nonsurgical orthopedic expansion especially in patients with narrow and high-arched palates.
Clinical example
Diagnosis and etiology
An 18-year-old healthy woman presented to the orthodontic clinic at the University of Wuhan for an orthodontic consultation [Figure 2]. The chief complaint was, “I have protruded appearance and my anterior teeth are not touching.” The medical history was non-contributory. Facial examination revealed a convex high-angle facial profile and an increased lower face height. The position of the mandible was retrognathic with a retrusive chin. The patient had a 5 mm of arch width discrepancy (measured by the distance of the alveolar crest between the first molars with the midpoint of the alveolus at the level of the cemento-enamel junction) accompanied by a unilateral crossbite on the left side. The mandibular dental midline deviated 2 mm to the left side. An occlusal examination revealed a Class III molar and canine relationship with a 4 mm anterior open bite and a skeletal Class II jaw relationship.
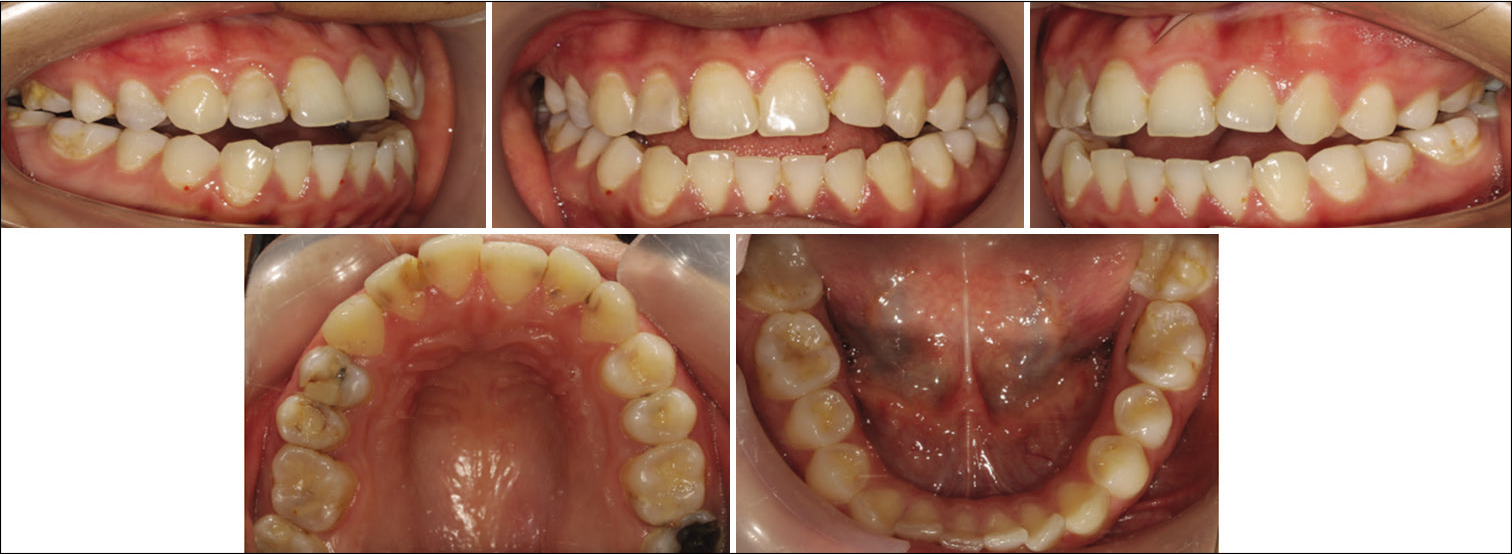
- Initial intraoral photographs showing patient with anterior open bite and posterior cross bites.
Treatment objective and alternatives
Possible treatment alternatives were discussed with the patient. A comprehensive orthodontic–orthognathic surgery treatment plan was selected to correct the problems in all three planes of space and macro- and mini-esthetics. The treatment objective was the orthopedic expansion to correct the posterior crossbite. This phase of treatment will be followed by comprehensive orthodontic treatment with fixed appliance and orthognathic surgery. In this case report, only the expansion phase of the treatment will be addressed to illustrate the fabrication of the custom-made MARPE appliance and demonstrate that following this treatment protocol can lead to good clinical results.
TREATMENT PROGRESS AND RESULTS
The patient consented to the treatment plan, and treatment was started with selection of the size of the MARPE expander according to the amount of skeletal expansion needed. A 10 mm MARPE appliance with four 2 × 11 mm microimplants inserted in 2 × 2 mm slots was selected. The insertion holes ensured a precision fit with the microimplants and secured perpendicular insertion of the microimplants. The 11-mm length was chosen by calculating the 2-mm height of the insertion slots, the 1–2 mm of gingival thickness, and 6–7 mm thickness between the two layers of palatal cortical bones. This was intended to promote bicortical engagement of the microimplants on the palate.
The activation of the expander started with three turns (1/4 mm/turn, 90° for each turn), followed by two daily activations. By the 2nd week, the patient reported hearing a click sound in the palatal suture region. This was followed by the appearance of an interincisal diastema the next day. [Figure 3] shows intraoral photography that confirms the suture opening and lack of collateral buccal inclination of maxillary molars after 35 activations [Figure 3a], after 20 months of retention [Figure 3b] and after debonding of custom-made MARPE appliance [Figure 3c]. The patient reported needing help with the activation because of increased mechanical resistance. Progress CBCT scans were taken after 5 days of completed expansion, 6 months and 20 months after maxillary expansion.

- Post-treatment intraoral photographs showing sutural opening confirmed by the presence of diastema between maxillary central incisors (a), after 20 months of retention (b) and after debonding of custom-made microimplant-assisted rapid palatal expansion appliance (c).
After completion of maxillary expansion, the upper craniofacial structures, including the maxillary basal bone, were noticeably widened, leading to complete elimination of the posterior crossbite. [Figure 4a] shows a relatively uniform increase in the width of the alveolar area and in the upper maxillofacial structures such as the zygoma and nasal bone [Figure 4a]. In the axial plane, the midpalatal suture was successfully split in almost parallel manner [Figure 4b]. The first molars and premolars showed slight increase in buccolingual angulation [Table 1 and Figure 4 c].
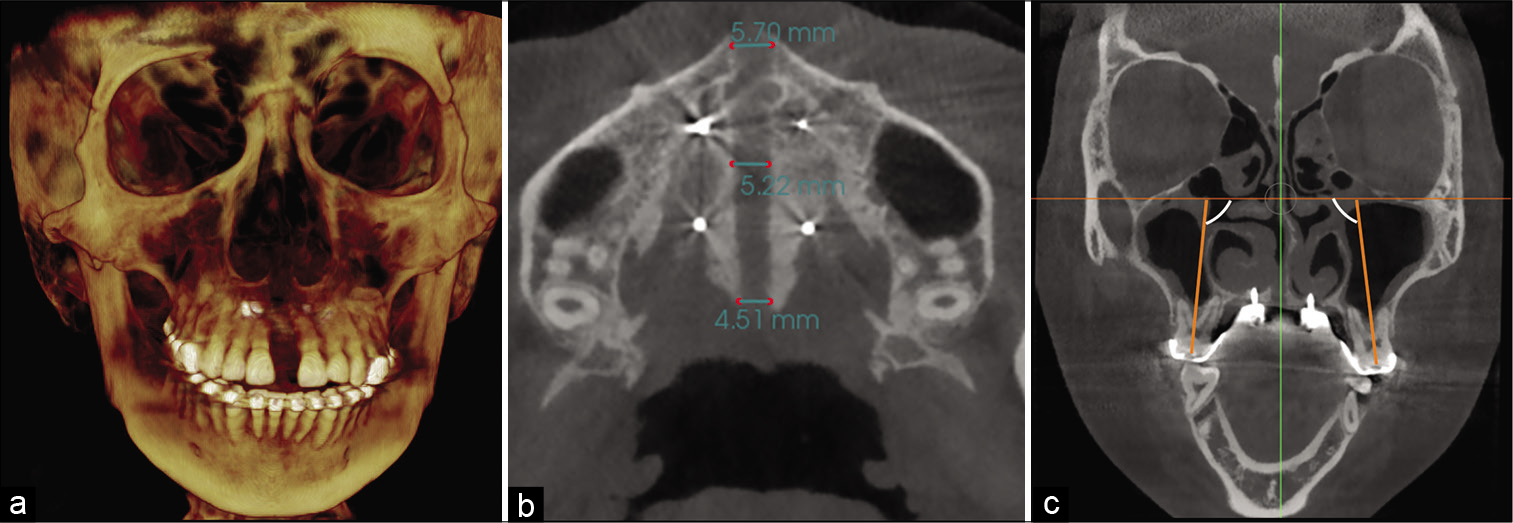
- Final frontal (a) and axial (b) Cone-beam computed tomography (CBCT) slices showing homogenous suture opening along the anterior and posterior regions and uniform separation of the hemimaxillae. (c) Frontal CBCT slice showing buccal tipping of posterior molar after maxillary expansion.
| Measurements | Before expansion | After expansion | Change of angulation compared with initial () |
|---|---|---|---|
| First premolars | 93.25 | 95.63 | 2.38 |
| Second premolars | 94.53 | 97.1 | 2.57 |
| First molars | 94.65 | 97.66 | 3.01 |
Significant increase in transverse skeletal width was found in intermolar distance, maxillary width at inferior palatine margin, nasal floor and at the lower interzygomatic distance as shown in [Table 2 and Figure 5a-c]. [Figure 6] shows the superimposition on pre-operative and directly post-operative three-dimensional CBCT expansion changes on coronal [Figure 6a] and axial [Figure 6b]; and superimposition of post-treatment and after 20 months of retention at the coronal [Figure 6c] and axial [Figure 6d] showing stability of skeletal changes.
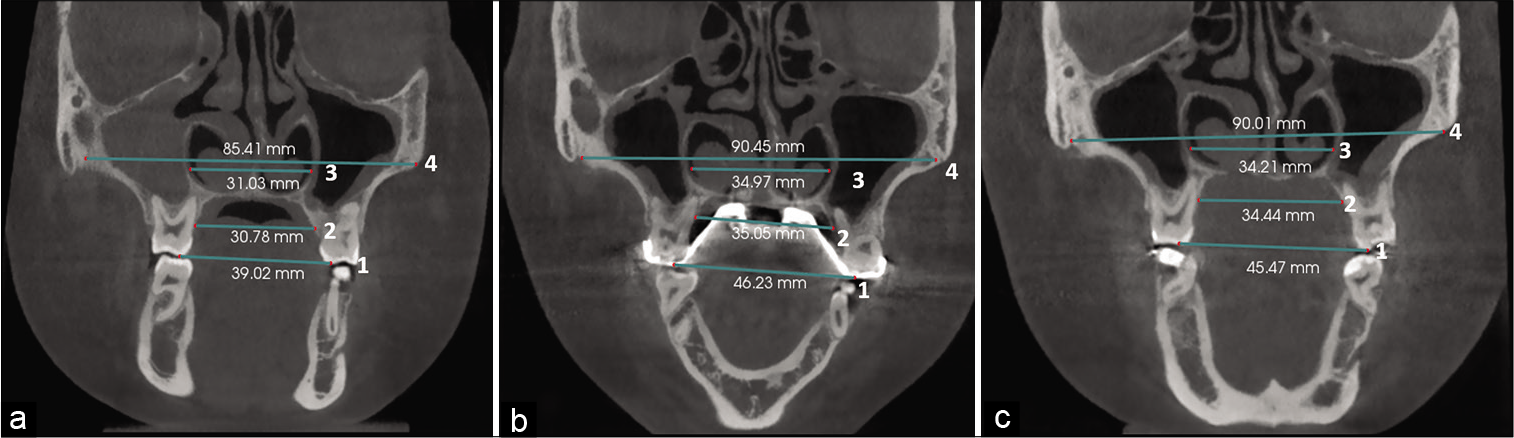
- Dental and skeletal linear measurements in the coronal zygomatic section: Maxillary width was measured at the level of intermolar distance (1), at the inferior palatine margin of alveolar process of maxilla (2), at the level of nasal floor (3), and maxillary width at the lower interzygomatic distance (4) in the pretreatment (a), directly after expansion (b), and after 20 months of retention (c).

- Superimposition of pre- and directly post-treatment cone-beam computed tomography scan at the coronal (a) and axial (b) section showing skeletal and dental changes with expansion, and superimposition of post-treatment and after 20 months of retention at the coronal (c) and axial (d) section showing stability of skeletal changes.
| Measurements | Before expansion (mm) | After expansion (mm) | After 20 months of retention (mm) |
|---|---|---|---|
| Intermolar distance | 39.02 | 46.23 | 45.47 |
| Inferior palatine margin of alveolar process of maxilla | 30.78 | 35.05 | 34.44 |
| Nasal cavity | 31.03 | 34.97 | 34.21 |
| Zygomatic bone | 85.41 | 90.45 | 90.01 |
The change in molar inclination was minimal due to the rigidity of the appliance and the force was designed to exert on the bone through the microimplants before the teeth were affected. A small change in buccal inclination was observed that may be related to the bodily rotation of the maxillary structure on each side. Buccal rotation or bending of the segments occurred throughout the arch, but the amount was greater from the posterior to anterior region[8] that may be the explanation to the changes of the molars and premolars.[5]
DISCUSSION
Different designs and technique in fabricating MARPE appliances can affect the clinical treatment outcome.[9] A MARPE appliance can be fabricated using only microimplant (palatal distractors) as anchorage or a hybrid design supported by both microimplant and teeth. The position in placing the jet screw as well as the placement of microimplants can also affect the orthopedic effect of the appliance. For patients who require treatment with MARPE appliances, four palatal mini-implants can be inserted to achieve bone-borne rapid palatal expansion, with two anterior mini-implants placed at the first premolar plane and two posterior ones placed at the first molar plane. From the standpoint of mini-implant stability, the posterior two mini-implants are susceptible to loosening and failure in adolescent female patients due to inadequate palatal bone support.[10] Thus, hybrid bone-borne and tooth-borne palatal expanders with four palatal mini-implants are recommended for adolescents who require treatment with MARPE appliances.
For custom-made maxillary skeletal expander, the casting base is designed according to the shape of the patient’s palate, close to the palatal mucosa. In this new design, one end consists of casting body, inserting hole and teeth retention device (circumferential rings) connected to another similar end by jackscrew expander. Connecting all parts in an integral casting unit increases the strength of the expansion appliance and avoids the distortion, deformation, or damage to the palatal mucosa because of increased resistance from the oromaxillofacial area. The custom-made maxillary rapid expander is fixed in the patient’s palate through the microimplant and the teeth casting band rings. Using the self-drilling microimplant placement method with appropriate digital key, the force can be effectively transmitted to the palate through the microimplant, opening of the midpalatal suture, and the maxilla.
The thickness of palatal soft tissues may influence an orthodontist’s decision on the length of mini-implants and affects biomechanical stability and overall success rates of mini-implants.[11] Thus, both palatal hard and soft tissue thickness should be considered in the planning of palatal mini-implant insertion. In our design, to ensure bicortical bone penetration, the microimplant length should be the sum of hard tissue thickness plus the soft tissue plus the height of the inserting hole and about 2–3 mm the depth of the nasal cavity.
A recent study by Lyu et al.[10] showed that the thicknesses of hard tissue and hard plus soft tissue at the first premolar plane were thickest among the four planes, followed by those at the second premolar molar, and second molar planes. In our design, the implant position is usually chosen between first premolar and first molar, 4–8 mm away from the midpalatal suture at the thickest part of palatal bone. Kang et al.[12] also reported that the bone thickness decreased laterally and posteriorly. It could be considered that this change was attributed to embryonic development. Development of the hard palate consists of the primary palate and the secondary palate. They fuse in the fetal development process and form the anterior and posterior palate.[13] Vertical thickening of the secondary palate is limited due to the rapid development of the tongue, so the thickness of the posterior palate is relatively thinner.
The anatomical shape of the palate, bone thickness of the implantation site, and bicortical penetration (oral and nasal -of the microimplants are determining factors for success of maxillary expansion with MARPE device. The current design incorporated several changes to increase the primary stability and provided a more efficient propagation of forces to the nasomaxillary complex. In our appliance fabrication, the casting base is designed according to the shape of the patient’s palate with equal diameter for both microimplant and inserting holes. The inserting hole is placed at the optimal implant position, parallel to palatal mucosa, guiding the microimplant to be vertically implanted into the palate. The inserting hole is tightly attached to the palatal mucosa with a height of about 1–2 mm and a diameter equal to that of microimplant diameter, generally 2 mm, that can ensure tight-fitting of microimplant preventing it from tilting and bending due to excessive lateral force during the expansion process. These changes added greater resistance to both the microimplant and jackscrew. It should be indicated in patients that higher resistance of the circummaxillary sutures is expected. Microimplant might be conceptualized to become part of orthodontic and orthopedic treatment carried out with elastics and wires and be useful as anchorage units.
Finally, with the advent of digital technology, intraoral scanners (IOS) are devices for capturing direct optical impressions in dentistry.[14] Optical impressions reduce patient discomfort; IOS are time-efficient and simplify clinical procedures for the dentist, eliminating plaster models and allowing better communication with the dental technician and with patients. In addition, 3D digital printing of pre-treatment study cast using software such as 3 Shape (Hangzhou lilac Medical Device LTD Company) can enable the best fit and accuracy of the custom-made appliances. The 3D stereolithographic file can then be sent directly to the dental laboratory and the patient-specific casting mold can be created (DUAL-150, laser melting [SLM] technology). This technique is cost effective other than the cost of the initial hardware. The files can be electronically transmitted to the laboratory for fabrication of the appliances. The turnaround time could be as little as 7 days before cementation in patient.
CONCLUSION
This paper described the procedure in fabricating a custom-made cast splint MARPE appliance that may overcome a few shortcomings of the commercially available appliances. Furthermore, the clinical example shows that fabrication of a cast splint MARPE appliance custom made to the patient’s anatomical need may result in better orthopedic separation of the palatal bones and satisfactory clinical results.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflict of interest
There are no conflict of interest.
References
- A longitudinal study on malocclusion in relation to signs and symptoms of cranio-mandibular disorders in children and adolescents. Eur J Orthod. 1990;12:399-407.
- [CrossRef] [PubMed] [Google Scholar]
- Occlusion, orthodontic treatment, and temporomandibular disorders: A review. J Orofac Pain. 1995;9:73-90.
- [Google Scholar]
- The transverse dimension: Diagnosis and relevance to functional occlusion. RWISO J. 2010;2:13-22.
- [Google Scholar]
- Microimplant-assisted rapid palatal expansion appliance to orthopedically correct transverse maxillary deficiency in an adult. Am J Orthod Dentofacial Orthop. 2016;149:716-28.
- [CrossRef] [PubMed] [Google Scholar]
- Transverse, vertical, and anteroposterior changes from bone-anchored maxillary expansion vs traditional rapid maxillary expansion: A randomized clinical trial. Am J Orthod Dentofacial Orthop. 2010;137:304.e1-12. discussion 304-5
- [CrossRef] [PubMed] [Google Scholar]
- Análise Microscópica da Sialometaplasia Necrosante e Apresentação de Uma Hipótese Sobre Suas Relações Etiopatogênicas, em Especial Com Aparelhos Disjuntores Palatinos São Paulo: Universidade de São Paulo; 2003.
- [Google Scholar]
- Facioskeletal and dental changes resulting from rapid maxillary expansion. Angle Orthod. 1966;36:152-64.
- [Google Scholar]
- Skeletal effects to the maxilla after rapid maxillary expansion assessed with cone-beam computed tomography. Am J Orthod Dentofacial Orthop. 2008;134:8-9.
- [CrossRef] [PubMed] [Google Scholar]
- Non-surgical treatment of transverse deficiency in adults using Microimplant-assisted rapid palatal expansion (MARPE) In: Dental Press J Orthod. Vol 22. 2017. p. :110-25.
- [CrossRef] [PubMed] [Google Scholar]
- Assessment of available sites for palatal orthodontic mini-implants through cone-beam computed tomography. Angle Orthod. 2020;90:516-23.
- [CrossRef] [PubMed] [Google Scholar]
- Recommended placement torque when tightening an orthodontic mini-implant. Clin Oral Implants Res. 2006;17:109-14.
- [CrossRef] [PubMed] [Google Scholar]
- Bone thickness of the palate for orthodontic mini-implant anchorage in adults. Am J Orthod Dentofacial Orthop. 2007;131(Suppl 4):S74-81.
- [CrossRef] [PubMed] [Google Scholar]
- Intraoral scanners in dentistry: A review of the current literature. BMC Oral Health. 2017;17:149.
- [CrossRef] [PubMed] [Google Scholar]






