Translate this page into:
Evaluation of white spot lesions around orthodontic brackets using different bonding agents – An in vivo study

*Corresponding author: Vaibhav Vashishta, Department of Orthodontics and Dentofacial Orthopedics, Sardar Patel Post Graduate Institute of Dental and Medical Sciences, Lucknow, Uttar Pradesh, India. drvaibhavvashishta@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Vashishta V, Kaul R, Singh A, Kapoor S, Grover S, Singhal M. Evaluation of white spot lesions around orthodontic brackets using different bonding agents – An in vivo study. APOS Trends Orthod 2021;11:309-16.
Abstract
Objectives:
The objectives of the study were to assess the white spot lesions around orthodontic bracket macroscopically using two different bonding agents – one with amorphous calcium phosphate (ACP) (Aegis Ortho) and one without ACP (Transbond XT).
Materials and Methods:
The study comprises 10 patients from 14 to 23 years of age. Patients were divided into control and study groups. Forty premolar teeth were then observed (20 teeth in each group). Bonding procedure was done and brackets were positioned on all four 1st pre-molars teeth and pre-treatment photographs were taken. The experimental material used was Aegis Ortho composite (study group) and Transbond XT (control group). Debonding procedure (by Wing deformation technique) was performed after 16 weeks which led to adhesive fracture at composite resin adhesive bracket interface leaving essentially all adhesive on the enamel. Then, follow-up photographs were taken to evaluate each.
Results:
Aegis Ortho containing ACP used for bonding purpose, provided lesser enamel demineralization than Transbond XT.
Conclusion:
Aegis Ortho for orthodontic bonding is significantly more beneficial and reliable, however, further investigations are also required to understand its clinical performance better.
Keywords
White spot lesions
Aegis Ortho
Transbond XT
Bonding
Debonding
Visual analog scale
INTRODUCTION
The term white spot lesion (WSL) was defined by Fejerskov et al. as “The first sign of carious lesion on enamel that can be detected with the naked eye.”[1,2] Unbalance between demineralization and remineralization of the enamel due to variations in enamel composition, local concentration of specific ions, organic material, and organic acids causes WSLs. The dissolution of the enamel is caused by organic acids produced by bacteria in the plaque. Demineralization in WSLs occurs mostly in the subsurface region of the enamel and can occur rapidly, as early as 4 weeks,[3] up to a depth of 75 μm. Fixed orthodontic appliances induce continued bacterial retention which increases the risk of caries development during orthodontic treatment. The design and surface characteristics of both orthodontic attachment and composite may influence plaque retention.[4] Furthermore, method of ligation of archwire is of importance in the development of WSLs.[5]
The reported incidence rates of WSL are 2–96% on the tooth surface.[6,7] The development of WSLs on the facial surfaces of anterior and posterior teeth represents an unesthetic side effect of orthodontic treatment that may counteract its beneficial results.
Several methods of white spot quantification can be used in a clinical setting but the ideal method should be simple, non-invasive, repeatable, reproducible, and accurate. Hence, methods like macroscopic evaluation can also be used, which include clinical examination, photographic examination, optical non-fluorescent methods, and optical fluorescent methods.
Visible WSLs can evoke concern from the patient. The treatment of WSLs, to produce a sound and esthetically pleasing enamel surface, is a paradox, yet unanswered. Aggressive and incisive techniques such as microabrasion and composite restoration have been used to resolve WSLs. Removal of stagnant plaque alone is not enough to achieve complete repair of WSL. Therefore, it is necessary to utilize secondary agents for repair during or post-orthodontic therapy.
During the past decade, Schumacher et al.[8] have been developing biologically active restorative materials that may stimulate repair of tooth structure through the release of caries fighting components including calcium and phosphorous, referred to as smart composites. They contain amorphous calcium phosphate (ACP) as bioactive filler material encapsulated in polymer binder. The anticariogenic potential of casein phosphopeptide ACP (CPP-ACP) has been demonstrated using laboratory, animal and human in situ models. Calcium and phosphate ions released from ACP materials, can be deposited on tooth structures as an apatite mineral which is similar to hydroxylapatite found in teeth.[9]
Hence, the main purpose of this in vivo macroscopic study was to compare the extent of white spot formation (demineralization) during the orthodontic treatment, by bonding the bracket onto tooth surface with two different bonding composites (Transbond XT and Aegis Ortho).
MATERIALS AND METHODS
Sample selection
A total number of 10 patients planned with all four first pre-molar extractions were selected (14–23 years of age), who had not undergone any previous dental or orthodontic treatment. The samples were divided into control group (Group A) and study group (Group B) in a randomized cross over arch design. Patients were not informed about which bonding composite was used on their teeth. Forty teeth (right and left maxillary and mandibular first premolars) were then evaluated, that is, 20 teeth/subjects in each group. Teeth were thoroughly evaluated for caries, restorations, dental structural abnormalities, or developmental defects. Full mouth oral prophylaxis was done, teeth were isolated with cheek retractor then a cellophane tape was spread over the premolar teeth and a hole corresponding to the bracket dimension was cut out to the required bracket attachment area to avoid the confusion of demineralization caused by etchant. A randomized cross arch design was selected for the study, for eliminating the bias/factors such as salivary flow rate, buffering capacity, and antimicrobial activity which can inhibit or reverse demineralization.
The bonding of four premolars, scheduled for extraction after 16 weeks, was done with Aegis orthodontic bonding light cure composite with ACP (AEGIS ORTHO The Harry J. Bosworth® Company, USA [Group B] and TRANSBOND XT 3 M UNITEK [Group A]). The bonding procedure was started with etching in the cut-out area of the tape [Figure 1]. During bonding, brackets were positioned on the appropriate teeth and excess material was removed with an explorer, the materials were then light cured. Both pre-treatment and follow-up photographs after 16 weeks of bonding were taken.

- Bonded teeth with etchant in the cut-out area of teeth for all first four pre-molars.
Examination of sample
Macroscopic examination was done by photographic evaluation of premolars using Sony DSLT α-58 camera with macro-objective lens and a ring flash. These standardized intraoral slides of pre and post bonded teeth at 90°, 20° above, and 20° below were evaluated. The images were cropped to include only premolars. Photographs were taken before bonding and at follow-up appointment 16 weeks later. Labial surface of tooth around orthodontic brackets was examined for the presence of WSLs [Figure 2]. Modified WSL index by Gorelick et al. was used to evaluate the teeth macroscopically.[10] The severity of WSL score is shown in [Figure 3].

- Pre-molars labial surface, pre and follow up appointment photographs at different angles.

- Severity of WSL score.
The WSL severity scores were
0 – No WSL formation
1 – Slight white spot or line formation
2 – Excessive white spot formation
3 – White spot formation with cavitation.
The severity of WSL (in visual analog scale [VAS]: 100 mm) was assessed by 10 observers. Hence, visual assessment of photographs was done by expert panel that comprised five postgraduate students of orthodontics department and five randomly selected individuals. In case of contradiction in results, the judgment of chief invigilators/postgraduate students would be considered as the final result. The overall mean WSL severity (VAS) of two groups further categorized as cavitation (0–25), severe (25–50), slight (50–75), and no WSL (75–100) and summarized in [Table 1] and also shown graphically in [Figure 4].
| Layman terminology | VAS scale | Gorelick et al. index | Dental professionals terminology |
|---|---|---|---|
| Very unattractive | 0–25 | 3 | Cavitation |
| Unattractive | 25–50 | 2 | Severe |
| Attractive | 50–75 | 1 | Slight |
| Very attractive | 75–100 | 0 | No WSL |
VAS: Visual analog scale, WSL: White spot lesion
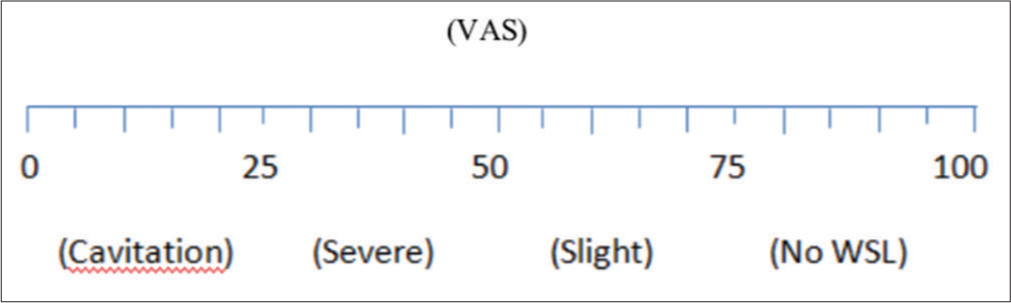
- Visual analog scale.
PowerPoint slides were projected on wall projector to maximally reduce the method error and increase the WSL identification.
RESULTS
Two independent groups were compared by Student’s t-test and Mann–Whitney U-test wherever applicable. The interobserver variability was compared by Kruskal– Wallis (W) one-way analysis of variance (ANOVA) and the significance of mean difference between the groups was done by Dunn’s test. The interobserver variability was assessed by Pearson correlation analysis. A two-sided (α = 2) P < 0.05 was considered statistically significant. All analyses were performed on STATISTICA software (Windows version 6.0).
Macroscopic evaluation
[Table 2] shows that the mean WSL severity in both groups increases (demineralization) at after debonding as compared to at before debonding and the demineralization was evident higher in Group A than Group B.
| Groups | Before bonding (n=20) |
After debonding (n=20) |
Change (after-before) |
W value | P value |
|---|---|---|---|---|---|
| Group A | 0.80±0.77 | 1.40±0.68 | 0.60±0.50 | 78.00 | 0.001 |
| Group B | 0.80±0.77 | 1.00±0.65 | 0.20±0.41 | 10.00 | 0.125 |
| U value | 200.00 | 136.00 | 120.00 | - | - |
| P value | 0.989 | 0.082 | 0.028 |
Comparing the mean WSL severity within the groups [Table 2], Wilcoxon test revealed significant increase in WSL severity after debonding as compared to before bonding in Group A (0.80 ± 0.77 vs. 1.40 ± 0.68, W = 78.00, P = 0.001); however, in Group B, it did not increase significantly after debonding as compared to before bonding (0.80 ± 0.77 vs. 1.00 ± 0.65, W = 10.00, P = 0.125).
Similarly, comparing the mean WSL severity between the groups [Table 2], the Mann–Whitney U-test revealed similar WSL severity between the two groups at both periods (before bonding: 0.80 ± 0.77 vs. 0.80 ± 0.77, U = 200.00, P = 1.000; after debonding: 1.40 ± 0.68 vs. 1.00 ± 0.65, U = 136.00, P = 0.082).
However, comparing the net mean change (after debonding-before bonding) in WSL severity of two groups [Table 2], Mann–Whitney U-test revealed significantly different and 23.0% higher demineralization in Group A as compared to Group B.
Modified Gorelick scale
The net mean change scores of WSL severity (score) of two groups further categorized as no WSL (0), slight (1), and severe (2) and summarized in [Table 3 and Figure 5]. At final evaluation, there were eight teeth with no WSL (no severity) (40.0%) and 12 with slight WSL (60.0%) in Group A while in Group B were 16 (80.0%) and 4 (20.0%), respectively. However, there was no severe WSL (0.0%) in both groups. Comparing the WSL severity of two groups, χ2 test revealed significantly different and 40.0% higher No WSL in Group B as compared to Group A (P = 0.010).
| WSL severity | Group A (%) (n=20) |
Group B (%) (n=20) |
χ2 value (DF=1) |
P value |
|---|---|---|---|---|
| No WSL | 8 (40.0) | 16 (80.0) | 6.67 | 0.010 |
| Slight | 12 (60.0) | 4 (20.0) | ||
| Severe | 0 (0.0) | 0 (0.0) |
WSL: White spot lesion

- Frequency distribution of WSL severity of two groups.
Further, there were less “slight” WSL found in the Group B when compared to Group A, which also that Group B is significantly more reliable.
VAS
The severity of WSL (in VAS: 100 mm) was assessed by five postgraduate students (P1, P2, P3, P4, and P5) and five laymen (L1, L2, L3, L4, and L5). The observed WSL severity of both Group A and Group B is summarized in [Figures 6 and 7], respectively. The observed mean WSL severity in Group A ranged from 47.85 mm to 51.75 mm with overall mean (± SD) 49.41 ± 16.55 mm. Comparing the observed mean WSL severity among different observers, Kruskal–Wallis ANOVA revealed similar observed WSL severity among the observers (F = 0.08, P = 1.000). Similarly, the observed mean WSL severity in Group B ranged from 65.50 mm to 68.50 mm with overall mean (± SD) 67.13 ± 17.62 mm. Comparing the observed mean WSL severity among different observers, Kruskal–Wallis ANOVA revealed similar observed WSL severity among the observers (F = 0.05, P = 1.000).

- Observed white spot lesion severity of Group A by different observers.

- Observed white spot lesion severity of Group B by different observers.
Further, to see the intraobserver reliability of severity of WSL, the observed WSL severity was correlated among the observers and summarized in [Tables 4 and 5]. The Pearson correlation analysis revealed significantly high correlation from 0.86 to 0.97 among the observers in both groups indicating high reliability among the observers.
| Observers | P1 | P2 | P3 | P4 | P5 | L1 | L2 | L3 | L4 | L5 |
|---|---|---|---|---|---|---|---|---|---|---|
| P1 | 1.00 | |||||||||
| P2 | 0.91 | 1.00 | ||||||||
| P3 | 0.97 | 0.92 | 1.00 | |||||||
| P4 | 0.93 | 0.86 | 0.93 | 1.00 | ||||||
| P5 | 0.92 | 0.92 | 0.88 | 0.87 | 1.00 | |||||
| L1 | 0.93 | 0.87 | 0.95 | 0.91 | 0.87 | 1.00 | ||||
| L2 | 0.93 | 0.88 | 0.94 | 0.92 | 0.90 | 0.87 | 1.00 | |||
| L3 | 0.94 | 0.88 | 0.92 | 0.89 | 0.89 | 0.88 | 0.87 | 1.00 | ||
| L4 | 0.95 | 0.89 | 0.96 | 0.90 | 0.88 | 0.93 | 0.96 | 0.88 | 1.00 | |
| L5 | 0.97 | 0.91 | 0.95 | 0.91 | 0.89 | 0.92 | 0.90 | 0.92 | 0.92 | 1.00 |
| Observers | P1 | P2 | P3 | P4 | P5 | L1 | L2 | L3 | L4 | L5 |
|---|---|---|---|---|---|---|---|---|---|---|
| P1 | 1.00 | |||||||||
| P2 | 0.94 | 1.00 | ||||||||
| P3 | 0.93 | 0.88 | 1.00 | |||||||
| P4 | 0.94 | 0.92 | 0.90 | 1.00 | ||||||
| P5 | 0.92 | 0.91 | 0.90 | 0.94 | 1.00 | |||||
| L1 | 0.92 | 0.92 | 0.86 | 0.92 | 0.92 | 1.00 | ||||
| L2 | 0.97 | 0.90 | 0.93 | 0.91 | 0.90 | 0.88 | 1.00 | |||
| L3 | 0.97 | 0.91 | 0.92 | 0.96 | 0.94 | 0.92 | 0.94 | 1.00 | ||
| L4 | 0.97 | 0.94 | 0.91 | 0.94 | 0.90 | 0.90 | 0.94 | 0.94 | 1.00 | |
| L5 | 0.97 | 0.93 | 0.92 | 0.94 | 0.95 | 0.92 | 0.95 | 0.96 | 0.95 | 1.00 |
Thus, comparing the overall observed mean WSL severity of two groups, Mann–Whitney U-test revealed significantly different and higher (26.4%) WSL severity of Group B as compared to Group A (P = 0.003), indicating less severity in Group B as compared to Group A [Figure 8].

- Overall observed mean white spot lesion severity of two groups.
The overall WSL severity (VAS) of two groups further categorized as cavitation (0–25), severe (25–50), and no WSL (75–100) and summarized in [Table 6]. At final evaluation of WSL, there were nine teeth with severe WSL (45.0%), 9 with slight (45.0%), and 2 with no WSL (10.0%) in Group A while in Group B, there were 3 (15.0%), 7 (35.0%), and 10 (50.0%), respectively. However, there was no WSL with cavitation (0.0%) in both groups. Comparing the WSL severity of two groups, χ2 test revealed significantly different and 40.0% higher no WSL in Group B as compared to Group A.
| WSL severity | Group A (%) (n=20) |
Group B (%) (n=20) |
χ2 value (DF=2) |
P value |
|---|---|---|---|---|
| Cavitation | 0 (4.0) | 0 (0.0) | 8.58 | 0.014 |
| Severe | 9 (45.0) | 3 (15.0) | ||
| Slight | 9 (45.0) | 7 (35.0) | ||
| No WSL | 2 (10.0) | 10 (50.0) |
WSL: White spot lesion
DISCUSSION
WSLs continue to remain a serious problem in orthodontics. Patients with fixed orthodontic appliances are vulnerable to plaque accumulation and consequently WSL formation around brackets in spite of conventional oral hygiene methods. Moreover, the clearance of bacterial plaque adjacent to the irregular surfaces of brackets, bands, wires, and other attachments by saliva and the cheeks is reduced.[1,3,11-13]
White spot is initiated through demineralization of tooth mineral by organic acids. Plaque bacteria, following exposure to fermentable carbohydrates, produce organic acids. When a critical pH of 5.5 is reached, the organic acids diffuse into the enamel surface through the acquired pellicle, initiating demineralization.[14,15] In vivo experiments by Ogaard et al.[16] have shown that visible WSLs can develop in orthodontic patients within 4 weeks in the absence of any fluoride supplementation.[12,17] Therefore, in case of highly vulnerable patients, the use of extra preventive and cautious measures is advised.
To produce an esthetically pleasing remineralized enamel, the demineralized enamel must be restored to a substrate and surface arrangement that will allow light to behave as it does in the surrounding, unaffected enamel.[18]
The proposed mechanism of anticariogenicity of CPP-ACP is that it acts as a calcium-phosphate reservoir. It buffers the activities of free calcium and phosphate ions in the plaque fluid at the tooth surface helping to maintain a state of supersaturation with respect to enamel mineral. The bioavailability of these remineralizing ions safeguards the enamel surface from acidogenic attack, thereby depressing enamel demineralization during these cariogenic challenges and enhancing the subsequent remineralization of the enamel.[19,20]
One of the benefits of this peptide compound is that it has been shown to incorporate well into the salivary pellicle, thereby inhibiting the adhesion of cariogenic bacteria, specifically Streptococcus sobrinus and Streptococcus mutans, to the tooth surface.[21] This technology has entered the orthodontic market place in the form of resin bracket bonding cement containing ACP complex.[22]
Further, many studies have reported greater prevalence of WSL on incisors as compared to other teeth.[12,23] According to Benson,[24] measurement of both incidence and severity depends on the method of recording the WSL. According to him, two main methods of recording WSL at macroscopic level are visual inspection and clinical photographs. Photographs have the advantage of providing a permanent record of the appearance of the tooth.[25]
For macroscopic evaluation of WSL, semi-quantitative classification system by Gorelick et al.[10] and its modification (1988) to simplify scoring was considered and teeth were evaluated before and during orthodontic treatment. The period of observation in the previous studies has ranged from up to 4 weeks of bonding during orthodontic treatment.[26,27]
The present study revealed significant increase in WSL severity after debonding as compared to before bonding in Group A but in Group B (study group), it did not increase significantly after debonding as compared to before bonding. This was in accordance with the study of Richter et al.[28]
However, in the present study, comparing the net mean change (after debonding-before bonding) in WSL severity of two groups, the study showed significantly different and 23.0% higher demineralization in Group A as compared to Group B.
On macroscopic examination, severity of WSL was highest in Group A than in Group B and in the present study while comparing the frequency (%) of WSL severity of both groups, significant difference was found between Group B as compared to Group A. This finding suggests that WSL formation in the control group is more as compared to Group B. The observed correlations were in agreement with those described by Enaia et al. and Ballard et al.[29,30]
Intergroup comparisons for before bonding and after debonding for both the control and experimental groups revealed a similar WSL severity between the two groups at both periods.
Comparing the observed mean WSL severity among different observers, the present study revealed similar observed WSL severity among the observers (F = 0.05, P = 1.000). The Pearson correlation analysis revealed significantly high correlation from 0.86 to 0.97 among the observers in both groups indicating high reliability among the observers.
On observing the overall observed mean WSL severity of two groups, the present study revealed significantly less WSL severity in Group B as compared to Group A. However, in another study, the effects of casein phosphopeptide amorphous calcium fluoride phosphate (CPP-ACFP) paste versus control paste on the remineralization of white spot caries lesions and on plaque composition were tested and a significant decrease in fluorescence loss was found with respect to baseline for both groups and no difference was found between groups.[31] Furthermore, the size of the lesion area did not change significantly overtime or between the groups. Hence, they observed no clinical advantage for use of the CPP-ACFP paste supplementary to normal oral hygiene over the time span of 12 weeks.
Comparing the WSL severity of two groups, the present study revealed significantly different and 40.0% higher no WSL in Group B as compared to Group A.
CONCLUSION
The present study concluded that ACP-containing orthodontic composite, Aegis Ortho provided higher reductions in enamel demineralization when compared with the control group. The use of Aegis Ortho for orthodontic bonding would appear to significantly prevent the development of enamel demineralization. Further investigations are needed to evaluate the clinical performance and to better understand any possible adverse effects of newly developed bonding materials from its use, either during or after orthodontic treatment.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Clinical and histological manifestation of dental caries In: Dental Caries: The Disease and Its Clinical Management. Vol 1. Copenhagen, Denmark: Blackwell Munksgaard; 2003. p. :71-99.
- [Google Scholar]
- Orthodontic treatment-related white spot lesions: A 14-year prospective quantitative follow-up, including bonding material assessment. Am J Orthod Dentofacial Orthop. 2010;138:136.e1-8. discussion 136-7
- [CrossRef] [PubMed] [Google Scholar]
- Orthodontic appliances and enamel demineralization Part 1 Lesion development. Am J Orthod Dentofacial Orthop. 1988;94:68-73.
- [CrossRef] [Google Scholar]
- Clinical comparison of direct versus indirect bonding with different bracket types and adhesives. Am J Orthod. 1978;74:62-78.
- [CrossRef] [Google Scholar]
- Ligature wires and elastomeric rings; two methods of ligation and their association with microbial colonization of Streptococcus mutans and lactobacilli European. J Orthod. 1991;13:416-20.
- [CrossRef] [PubMed] [Google Scholar]
- Decalcification during orthodontic treatment with fixed appliances--an overview. Br J Orthod. 1992;19:199-205.
- [CrossRef] [PubMed] [Google Scholar]
- Caries-inhibiting effect of preventive measures during orthodontic treatment with fixed appliances. A systematic review. Caries Res. 2004;38:413-20.
- [CrossRef] [PubMed] [Google Scholar]
- The use of amorphous calcium phosphate composites as bioactive basing materials, their effect on the strength of the composite / adhesive /dentin bond. J Am Dent Assoc. 2007;138:1476-84.
- [CrossRef] [PubMed] [Google Scholar]
- Quantitive assessment of the efficacy of amorphous calcium phosphate/methacrylate composites in remineralizing caries-like lesions artificially produced in bovine enamel. J Dent Res. 1996;75:1679-86.
- [CrossRef] [PubMed] [Google Scholar]
- Incidence of white spot formation after bonding and banding. Am J Orthod Dentofacial Orthop. 1982;81:93-8.
- [CrossRef] [Google Scholar]
- Enamel demineralization following orthodontic treatment. Am J Orthod Dentofacial Orthop. 1982;82:62-7.
- [CrossRef] [Google Scholar]
- The effect of a fluoride program on white spot formation during orthodontic treatment. Am J Orthod Dentofacial Orthop. 1988;93:29-37.
- [CrossRef] [Google Scholar]
- Caries lesions after orthodontic treatment followed by quantitative light-induced fluorescence: A 2-year follow-up. Eur J Orthod. 2007;29:294-8.
- [CrossRef] [PubMed] [Google Scholar]
- White spot lesions: Formation, prevention, and treatment. Semin Orthod. 2008;14:174-182.
- [CrossRef] [Google Scholar]
- Determinants of enamel decalcification during simulated orthodontic treatment. Angle Orthod. 2011;81:836-42.
- [CrossRef] [PubMed] [Google Scholar]
- Prevalence of white spot lesions in 19 year olds: A study of untreated and orthodontically treated persons 5 years after treatment. Am J Orthod Dentofacial Orthop. 1989;96:423-7.
- [CrossRef] [Google Scholar]
- Effect of fluoride varnish on demineralization adjacent to brackets bonded with RMGI cement. Am J Orthod Dentofacial Orthop. 2002;122:125-34.
- [CrossRef] [PubMed] [Google Scholar]
- In-vitro study of the effect of casein phosphopeptide amorphous calcium fluoride phosphate on iatrogenic damage to enamel during orthodontic adhesive removal. Am J Orthod Dentofacial Orthop. 2011;139:543-51.
- [CrossRef] [PubMed] [Google Scholar]
- Prevention of white spot lesions in orthodontic practice: A contemporary review TR. Aust Dent J. 2006;51:284-9.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of the fluoride gels and varnishes comparing to CPP-ACP complex on human enamel demineralization/ remineralization. Actastomatol Croat. 2012;47:99-110.
- [CrossRef] [Google Scholar]
- Incorporation of caseinoglycolmacropeptide and caseinophosphopeptide into the salivary pellicle inhibits adherence of mutans streptococci. J Dent Res. 1996;75:1779-88.
- [CrossRef] [PubMed] [Google Scholar]
- Amorphous calcium phosphate and its application in dentistry Zhao et al. Chem Cent J. 2011;5:40.
- [CrossRef] [PubMed] [Google Scholar]
- Risk factors for incidence and severity of whitespot lesions during treatment with fixed orthodontic appliances. Am J Orthod Dentofacial Orthop. 2010;138:188-94.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of white spot lesions on teeth with orthodontic brackets. Semin Orthod. 2008;14:200-8.
- [CrossRef] [Google Scholar]
- Durability of esthetic improvement following icon resin infiltration of multibracket-induced white spot lesions compared with no therapy over 6 months: A single-center, split-mouth, randomized clinical trial. Am J Orthod Dentofacial Orthop. 2013;144:86-96.
- [CrossRef] [PubMed] [Google Scholar]
- In vivo inhibition of demineralization around orthodontic brackets. Am J Orthod Dentofacial Orthop. 2003;123:10-4.
- [CrossRef] [PubMed] [Google Scholar]
- Development of white spot lesions during orthodontic treatment: Perceptions of patients, parents, orthodontists, and general dentists. Am J Orthod Dentofacial Orthop. 2012;141:337-44.
- [CrossRef] [PubMed] [Google Scholar]
- Incidence of caries lesions among patients treated with comprehensive orthodontics. Am J Orthod Dentofacial Orthop. 2011;139:657-64.
- [CrossRef] [PubMed] [Google Scholar]
- White-spot lesions during multibracket appliance treatment: A challenge for clinical excellence. Am J Orthod Dentofacial Orthop. 2011;140:E17-24.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of 3 commercially available materials for resolution of white spot lesions. Am J Orthod Dentofacial Orthop. 2013;143:S78-84.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of casein phosphopeptide amorphous calcium fluoride phosphate paste on white spot lesions and dental plaque after orthodontic treatment: A 3-month follow-up. Eur J Oral Sci. 2010;118:610-7.
- [CrossRef] [PubMed] [Google Scholar]






