Translate this page into:
Gingival crevicular fluid alkaline phosphatase activity as growth marker: A systematic review

*Corresponding author: Dr. Siddharth Sonwane, Ph.D Student & Associate Professor, Department of Orthodontics, Government Dental College, Nagpur, Maharashtra, India. siddharth5678@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Sonwane S, Bhad W. Gingival crevicular fluid alkaline phosphatase activity as growth marker: A systematic review. APOS Trends Orthod 2022;12:131-8.
Abstract
Background:
This review synthesizes the available evidence about the individual skeletal maturity with biological maturity indicators and compares it with the levels of gingival crevicular fluid (GCF) alkaline phosphatase (ALP) activity in growing children.
Aims:
This systematic review aimed to clarify the question: Is GCF ALP a reliable biomarker to assess skeletal maturity during growth?
Objectives:
The objective of this systematic review is to collect, compile, and review the existing evidence on the levels of GCF ALP activity in growing children and comparing its reliability with contemporary growth indicators.
Materials and Methods:
A literature appraisal executed using Entres PubMed, www.ncbi.nim.nih.gov, Scupose, Hinary, Ebsco, Embass, Cochrane; Google Scholar Electronic database search engines were used. The MeSH term used “growth markers in gingival crevicular fluid” or “growth markers in growing children.” Studies published till October, 2020, were included in this study.
Data Extraction and Quality Assessment:
The data have extracted from the selected articles based on year of publication, study design, age of subjects, instruments used, and author’s conclusions. The quality assessment was executed using BIOCROSS Scale. This scale is exclusive for cross-sectional studies with biomarkers.
Results:
Literature search identified 731 records from electronic databases and from the partial grey literature (Google scholar) search. Finally, six articles fulfilled eligibility criteria included in the review.
Conclusions:
All the six studies concluded that GCF ALP activity is a reliable method in determining a skeletal maturity indicator in growing children.
Keywords
Biomarkers
Skeletal maturity indicators
Alkaline phosphatase
Gingival crevicular fluid
Growth markers
INTRODUCTION
Skeletal age is essential in formulating viable orthodontic treatment plans.[1] It is used to assess the stages of maturation, as the individual posses’ variation in timing, duration, and velocity of growth.[2] A skeletal maturity indicator helps in evaluating stage of maturation.[3]
The chronological age has a high correlation with skeletal maturity in average patients, but it is not applicable for an entire population as body mass index, ethnic and metabolic factors may influence an early or late skeletal development.[1-3] The most often accepted ways of predicting the skeletal maturation is the hand and wrist ossification method (Bowden).[4] A major disadvantage of this method is that patients need to be exposed to additional radiation (Zhao et al. 2012).[5] To avoid radiation exposure, Lamprasky[6] in 1972 proposed a cervical vertebral maturation assessment technique. This technique avoids additional radiation exposure as it is assessed on lateral cephalometric radiographs, but the limitation is its a subjective method (Nestman et al. 2011).[7]
Considering drawbacks of the previous studies’ researchers have proposed that a skeletal maturity is an integral part of cells, molecules, genes, enzymes, and hormones, which can be assessed in the body to predict its incidence of outcomes and are known as biochemical marker or biomarkers.[1-7] They can be measured in body fluids as blood, saliva, and urine. Juul (1994) was first to measure serum insulin-like growth factor-I (IGF-1) and reported it as a skeletal maturity indicator.[8] In 1977, Fleisher et al. reported that in serum the levels of bone specific alkaline phosphatase (ALP) increases and the levels reduces during pre- adolescence and adult age.[9] Hence may be considered to be a potential marker for assessment of skeletal growth. The major limitations of these serum studies are its an invasive method hence difficult to get ethical clearance as the subjects are left untreated after sample collection.[6-8]
To overcome the drawbacks of serum biomarkers, Hizuka et al. showed that the presence of insulin-like growth factor-I in urine and hypothesized that the quantity of IGF-1 in urine may vary in subjects depending on either growth hormone excess or growth hormone deficiency.[10] Although, it is a non-invasive procedure; it evaluates the status of overall (systemic) bone activity, the data are still scarce and insufficient to deploy urine IGF-I as biomarkers for precise estimation of growth.[11]
The oral fluids as saliva can offers patient-specific diagnostic test for skeletal maturation as it is composed of local and systemically derived biomarkers (Alfano, 1974).[12] This method is most commonly preferred because of ease of sample collection, safe, and non-invasive. Pellegrini in 2008 investigated and showed that salivary IGF-I as biomarker and hypothesized that its levels varies with respect to pubertal stages in healthy growing children.[13] However, the limitations of saliva study are it contaminates the molecule of identification, as saliva has a wide spectrum of components such as proteins/peptides, nucleic acids, electrolytes, and hormones in it, and poor repeatability due to diurnal variations in flow (Silva et al. 2013).[14]
In healthy periodontium, gingival crevicular fluid (GCF) is derived from serum and can subsequently be used to investigate subclinical alterations in tissue metabolism and bone remodeling (Armitage, 2004).[15] Looks and Tjoa. (2005)[16] identified eight potential biomarkersin GCF as ALP, β glucuronidase, cathepsin B, collagenase-2 (matrix metalloproteinase, MMP-8), gelatinase (MMP-9), dipeptidyl peptidase II and III, and elastase, among them ALP is most precious as it specify local and systemic bone remodeling. ALP is a membrane-bound glycoprotein involved in maintenance of alveolar bone and renewal of the periodontal ligament.[15,16]
Highlighting the bone specific property of ALP, Perinitti (2012) showed age related variations in the levels of GCF ALP activity and compared it with cervical vertebral maturation stages and hypothesized those levels of GCF ALP activity are reliable skeletal maturity indicators.[17] Furthermore, in 2017 Perinetti et al. reported that the repeatability of enzyme alkaline phosphates was more acceptable than cervical vertebral maturity indicators.[18] Therefore, GCF ALP activity and its levels can offer a potential specific status.[1-18]
Objective
The objective of this systematic review is to collect, compile, and review the existing evidence on use of GCF ALP activity in growing children and comparing its reliability with existing growth indicators.
Focused question
Is GCF ALP a reliable biomarker to assess skeletal maturity during growth?
MATERIAL AND METHODS
In this systematic review the above question was framed using Population, Intervention, Comparison, Outcomes format.
Population
Periodontally healthy and growing children with the mean age group 13.5 (range of 8–18 years).
Intervention
Observing and analyzing the levels of GCF ALP activity during growth.
Comparison
Each levels of ALP were comparing with available individual skeletal maturity ondicators.
Outcomes
Levels of ALP activity at each stage of individual skeletal maturity indicator.
Information sources
The search approach for this systematic review was to assess the levels of gingival crevicular fluid ALP activity and compare it with various skeletal maturity indicators; a comprehensive literature search was carried out from distinct internet sources. The following search engine sources of data were used in the search of suitable articles fulfilling the study purpose: The Medline database (Entrez PubMed, www.ncbi. nim.nih.gov), Scopus, Hinary, Ebsco, Science direct, Google scholar, Jgate, Helinet and manual grey literature search using GDCH College Nagpur and departmental library sources were conducted. An additional source of data was generated by screening all cross reference lists from selected articles that could meet the eligibility criteria of the study. The search covered from January 1970 to December 2019 for articles written and published in English. The keywords used to identify appropriate articles are illustrated in [Table 1].
| S. No. | Primary key word | Secondary key words |
|---|---|---|
| 1. | Gingival crevicular fluid alkaline phosphatase activity as skeletal maturity indicator | Quality assessment of gingival crevicular fluid alkaline phosphatase activity in orthodontic diagnosis |
| 2. | Gingival crevicular fluid alkaline phosphatase activity versus skeletal maturity indicators | Growth assessment using gingival crevicular fluid alkaline phosphatase activity in growing children |
| 3. | Repeatability of gingival crevicular fluid alkaline phosphatase activity in comparison with other skeletal maturity indicators | Repeatability of gingival crevicular fluid biomarkers as skeletal maturity indicator |
Selection criteria
The selection strategy for each article is based on inclusion and exclusion criteria illustrated in [Table 2]. The articles included for this systematic review are RCTs, meta-analyses, descriptive, diagnostic, cross-sectional, and longitudinal studies published on gingival crevicular fluid as diagnostic tool in orthodontics. The data retrieved from these studies were assessed using Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) analyzer and the studies reporting on GCF ALP as a skeletal maturity indicator in growing children were selected.
| Inclusion criteria | Exclusion criteria |
|---|---|
| Meta-analyses on growth markers in GCF. Randomized control trials on growth markers and alkaline phosphates. Descriptive, diagnostic, cross-sectional, and longitudinal studies Articles in English Articles published from January 1970 to December 2019. Studies of skeletal maturity indicators with biomarkers and lateral cephalograms. |
Case reports, case series and letters to editor, opinion articles, and abstracts. Studies included BALP in growing children. Studies not involving alkaline phosphates from GCF Studies on GCF ALP in inflammatory condition and on other than growth indicators. Studies on GCF ALP for tooth movement and alveolar bone remodeling. Studies on adults on GCF biomarkers. |
Data collection
A series of electronic databases were explored using various search strategies including the above-mentioned keywords and their permutation to identify appropriate data. Only cross-sectional studies were found providing clear information about the levels of GCF ALP activity in each stage of pubertal growth. The number of studies identified was 731 and 03 through electronic database search and manual hand-search, respectively. After meticulous analyzing the titles, 627 studies were excluded and 107 articles titles found appropriate. The titles of 107 studies were screened for eligibility in which 98 studies removed as duplicate thus; only nine articles progressively met the eligibility criteria. The obtained nine articles were concealed for inclusion and three articles were removed in which two articles were reviews of literature and one was blood serum study. Finally six articles were included for this systematic review [Figure 1]. Full texts six studies endow with clear full text information about levels of GCF ALP activity. The allocation of the journals in which these studies are published is tabulated in [Table 3].
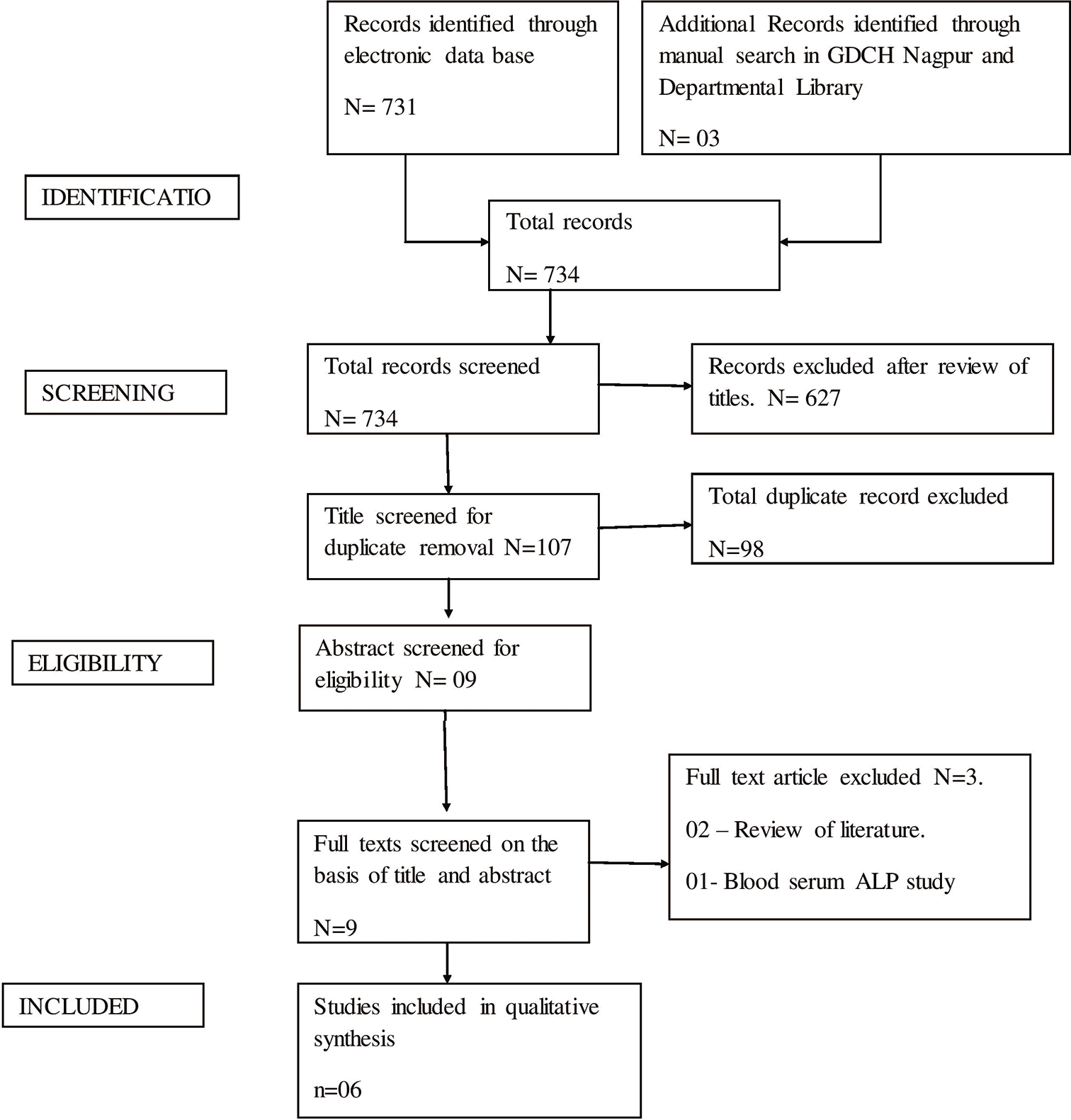
- PRISMA flow diagram included and excluded record.
| S. No. | Journal name | Author name | Year of publication |
|---|---|---|---|
| 1. | Biomedical and Pharmacology Journal 2015 | Arif Yezdani et al. | 2015 |
| 2. | Orthod Craniofac Res 2011 | Perinetti et al. | 2011 |
| 3. | South European Journal of Orthodontics and Dentofacial Research April 2016 | Perinetti et al. | 2016 |
| 4. | Angle Orthodontist | Franchi et al. | 2012 |
| 5. | Int J Res Med. 2018 | Patil | 2018 |
| 6. | Progress in orthodontics | Perinetti et al. | 2011 |
Quality assessment
The quality assessment of the procedural accuracy of each article was executed as per the method described in BIOCROSS evaluation tool (Jan Wirsching et al. 2018).[19] The BIOCROSS evaluation tool was applied to assess the quality of each of biomarker cross-sectional studies. The studies were assessed and were designated “stars” on the basis of design, sample size and prior-estimate of sample size, withdrawals (dropouts), method error analysis, blinding in measurements, and adequate statistics. Checklist Provided By the Clarity Group at Mcmaster University to Evaluate the Risk of Bias to Assess the Quality Of cross-sectional studies.
RESULTS
Study selection
The search approach resulted in 734 articles across different medical and dental journals, out of which 731 and three (department library) articles were found through online and manual hand search, respectively. 107 articles were shortlisted. After meticulous reviewing of abstracts 98 titles were removed as duplicate, six full text articles were acquired to verify their acceptance with the eligibility criteria and eventually six articles satisfied the selection criteria which were selected for qualitative concoction of the systematic review. All the six authors were requested through email to share data but none of them responded. The obtained data from published articles were included and this comprised 66.6% of the total articles gathered succeeding to the data search. This systematic review was performed and report following the PRISMA, the outline of which is shown in [Figure 1].
Study characteristics
The study characteristics are reviewed in [Table 4]. The various biological growth markers such as hand wrist radiograph, chronologic age, and cervical vertebral maturity indicators were included to identify the skeletal maturity in growing children, in six studies different biological growth markers and biomarkers were employed to identify skeletal growth. The study characteristics assessment was carried out using BIOCROSS cross-sectional biomarker study evaluation tool and studies were categorized. Three studies compared GCF ALP with hand wrist radiograph; one study compared with chronological age and two studies compared with cervical vertebral maturity indicator.
| Issues to consider (IC) | Study | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Perinetti et al. | Perinetti et al. | Franchi et al. | Arif et al. | Perinetti et al. | Patil | |||||||||||||
| 0 | 1 | 2 | 0 | 1 | 2 | 0 | 1 | 2 | 0 | 1 | 2 | 0 | 1 | 2 | 0 | 1 | 2 | |
| 1) Hypothesis/Objective | * | * | * | * | * | |||||||||||||
| 2) Study population selection | * | * | * | * | * | * | ||||||||||||
| 3) Study population representativeness | * | * | * | * | * | * | ||||||||||||
| 4) Study population characteristics | * | * | * | * | * | * | ||||||||||||
| 5) Statistical analyses | * | * | * | * | * | * | ||||||||||||
| 6) Interpretation and evaluation of results | * | * | * | * | * | * | ||||||||||||
| 7) Study limitations | * | * | * | * | * | * | ||||||||||||
| 8) Specimen characteristic, assay methods | * | * | * | * | * | * | ||||||||||||
| 9) Laboratory measurement | * | * | * | * | * | * | ||||||||||||
| 10) Biomarker data modeling. | * | * | * | * | * | * | ||||||||||||
0: Not Mentioned, 1: Partially Mentioned, 2: Completely Mentioned
The procedural assessment of the quality of the selected studies was reviewed using the BIOCROSS cross-sectional biomarker study evaluation tool. The scores given were between 0, 1, and 2, indicating that the quality of the studies was low, moderate, and high, respectively. The comprehensive quality appraisal of all studies is illustrated in [Table 5].
| Issue consideration | Hypothesis/ objective | Sample selection | Cofounder | Statistical analysis | Interpretation and evaluation of results | Study limitations | Specimen characteristics and assay methods | Laboratory measurement | Biomarker data modeling |
|---|---|---|---|---|---|---|---|---|---|
| Perinetti et al., 2011 | 2 | 1 | 0 | 2 | 1 | 2 | 2 | 2 | 1 |
| Perinetti et al., 2011 | 2 | 2 | 1 | 2 | 2 | 2 | 1 | 1 | 2 |
| Franchi et al., 2012 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 1 |
| Arif Yezdani et al. 2015 | 1 | 2 | 0 | 1 | 1 | 0 | 1 | 0 | 1 |
| Perinetti et al., 2016 | 2 | 2 | 2 | 2 | 2 | 2 | 1 | 1 | 2 |
| Patil, 2018 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 2 | 1 |
Risk of bias assessment
The results of risk of bias assessment within studies are presented using Checklist Provided By the Clarity Group at Mcmaster University for cross-sectional studies and are illustrated in [Table 6]. Four of the six included cross-sectional studies satisfactorily addressed the method of stratified random sampling. The rest of two studies did not disclose sampling procedure. For blinding, in these studies only detection bias were possible and performance bias not possible as subjects and operators were well explained about the clinical procedures. The detection blinding was performed in all the studies.
| Domains Assessed for Quality Evaluation | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Is the source population representative of the population of interest? | Is the response rate adequate? | Is there little missing data? | Is the survey clinically sensible? | Is there any evidence for the reliability and validity of the survey instrument | ||||||||||||||||
| A | B | C | D | A | B | C | D | A | B | C | D | A | B | C | D | A | B | C | D | |
| Arif Yezdani et al. | * | * | * | * | * | |||||||||||||||
| Perinetti et al. | * | * | * | * | * | |||||||||||||||
| Perinetti et al. | * | * | * | * | * | |||||||||||||||
| Franchi et al. | * | * | * | * | * | |||||||||||||||
| Patil | * | * | * | * | * | |||||||||||||||
| Perinetti et al. | * | * | * | * | * | |||||||||||||||
A: Definitely yes (low risk of bias), B: Probably yes, C: Probably no, D: Definitely no (high risk of bias)
Synthesis of results
One out of the six studies investigated levels of GCF ALP activity and compared with hand wrist radiographic method in growing children with little variation in measurement of ALP assay. One study investigated the levels of GCF ALP activity and compared it with chronologic age. Two studies by Perinitti assessed levels of GCF ALP activity with cervical vetibral maturity indicators and these carried out a multiple regression study to demonstrate and compare the variation in the levels of GCF ALP activity between age and gender in growing children.[20]
DISCUSSION
Determination of individual skeletal maturity is beneficial to both orthodontist and the patient.[1-20] Numerous maturity indicators are available based on morphologic and radiographic methods. Lateral cephalometric radiographic method as cervical vertebral maturation staging is preferred and adopted by the American Board of Orthodontics over the hand-wrist radiographic method to avoid extra radiation exposure.[21] Although, cervical vertebral maturation index (CVMI) is the contemporary method but radiation exposures and variability in subjective assessment affects its reliability.[22]
Identifying morphologic stages of bone are mainly under the influence of local factors but the developing bone is under influence of both local and systemic factors.[23] Thus, biomarkers have provided a new possibility to represent the agents who are involved in direct bone growth and remodeling.[24] Furthermore, biochemical marker or biomarkers can be measured in body fluids as blood (serum), saliva, and urine.[25] Serum biomarker getting ethical clearance is the issue, urine biomarker is difficult as for children to hold secondary urine sample and in saliva as its composition contaminates the identifying molecule has limitations.[21-25]
To date, Perinetti et al., Gussepe, Patil, Franc Hi, and other researchers have conducted studies to determine the level of GCF ALP activity with available contemporary skeletal maturity indicators. These studies have reported that levels of GCF ALP activity varies with individual skeletal maturity stage.[17,18,20-22] Thus, the objective of this review was to collect, compile, and review, all the existing data allied to the levels of GCF ALP activity and individual skeletal maturity indicators.
Summary of evidence
Study conducted by Perinetti et al. (2010)[17] aimed to compare the levels of enzyme GCF ALP activity and cervical vertebral maturity phase in growing children. This study employed the use of a spectrophotometer to evaluate GCF ALP levels and a lateral cephalogram for cervical vertebral assessment. A sum of 74 subjects participated with mean age of 13.5 years (8 years to 18 years) and study designed was prospective cross-sectional. The selected subjects were sent for professional scaling 1 week before sample collection and also kept on oral mouth rinse. On the day of sample collection a periodontal status was assessed (pocket depth was <3 mm, bleeding on probing). GCF collected from maxillary central incisors using periostrips were sent for analysis. The obtained result was recorded as per pubertal growth spurts of the subjects. Mean levels of GCF ALP activity at pre-pubertal stage was 49.7 IU, pubertal stage 87.5 IU, and at post-pubertal stage 45.7 IU.
This study has a low risk bias and is medium to high quality study as the researcher have clearly mentioned the research hypothesis, sampling methods, and biomarker assay [Tables 5 and 6]. However, cofounding factor and background factor have not been illustrated.
One year later Perinetti et al. (2011)[17] made an attempt to compare the level of GCF ALP activity with chronologic age. In this study, level of GCF ALP activity was evaluated using colorimeter. A sum of 87 patients with mean age group of 13.5 years (8–18 years) and distribution of sample was made using a stratified random sampling method. Before GCF collection patients were sent for professional scaling followed by one mouth rinses. One week later, sample was collected and lateral cephalogram was taken. The obtained level of GCF ALP activity was arranged as per growth spurts [Tables 5 and 6]. The steep rise and immediate fall in the levels of enzyme activity were seen at pubertal and post pubertal stages, respectively.
The overall this study has low risk and high quality as they have mentioned the research hypothesis, sampling methods, biomarker assay [Table 6], and also clarified about factors affecting GCF ALP levels.
Patil[21] performed a study to compare the level of GCF ALP activity with Bjork, Grave, and Brown hand wrist radiographic method. In their study, nine radiographic stages were condensed into three stages as SMIs 1 to 3 phase one (pre-pubertal phase), SMIs 4 to 5 second phase (pubertal phase), and SMIs 6 to 9 third phase (post-pubertal phase). Study sample were 65 with mean age of 13.6 years (range of 8–17 years). Volunteers and registered patients were sent for professional scaling and 1 week later sample was collected using pipit and ALP activity was recorded using spectrophotometer. The result is arranged according to pubertal growth spurts.
The study has an overall high risk bias and is lower to medium quality as the researcher have used pipit for sample collection in which dead cells and macrophage sensitive to ALP reagent can produce false positive result. The author has not mentioned about sample distribution, factors affecting, and biochemical assay clearly [Tables 5 and 6].
In 2016, Perinetti et al.[18] conducted a cross-sectional prospective study with 100 patients of mean age 13.5 years (range of 8–17 years). The study was aimed to determine the level of GCF ALP activity and compare it with CVMI stage. The simple stratified method was used sample distribution. Registered patients were sent for professional scaling; later examination of periodontal condition, history of health drink intake was made. GCF collected with periostrips and ALP activity was analyzed [Table 6]. In this study, the diurnal variations in flow of GCF on ALP activity were investigated by collecting sample at 10 am, 12 am and 3:30 pm; however, the flow was reduced but the levels of ALP activity remained similar throughout.
In this prospective cross-sectional study, the sample selection, distribution, and repeatability of levels of GCF ALP activity have been described clearly. Furthermor, the confounding and background factors were determined as it has influence on level of GCF ALP. In addition, appropriate analysis was done to estimate the level of GCF ALP and compared it with most accepted contemporary CVMI method. This research contributes toward low risk bias and high quality study.
Franchi et al. (2012)[22] conducted a cross-sectional study to evaluate and compare the levels of GCF ALP activity with different stages of hand wrist radiographs. The study included 78 growing subjects with a mean age of 13.5 ± 0.6 years. After the following of professional scaling of registered subject sample was collected and sent for laboratory. The obtained results are shown in [Table 6]. Overall, this study was a high risk and low quality study.
The study limitations
Only six studies have met the inclusion criteria in which two studies were high risk bias and low quality. All the six studies are cross-sectional with heterogeneity in their results due to use of different armamentarium. Perinitti, Patil studies have explained influences of confounding factor and back ground factors in results.
The study strength
The quality assessment and risk bias assessment evaluated using BIOCROSS scale. It is (BIOCROSS scale) exclusively for cross-sectional studies with biomarkers thus, reviewed studies’ results are reliable and reproducible.
Among included studies four studies are high quality with low risk bias and two low quality and high risk studies.
CONCLUSION
This systematic review was conducted to answer the question: “Is the GCF ALP enzyme activity reliable in determining skeletal maturity indicator in growing children?
Based on the examination of six retrieved studies, we have concluded that:
The quality standard of these studies ranged from low to medium/high. Four studies were Medium/high quality with low risk bias and two were low quality with high risk bias.
All the studies have reported that GCF ALP activity can be considered as non-invasive and reliable method as skeletal maturity indicator.
The contemporary skeletal maturity indicator (CVMI, Hand wrist, etc.) predict accurate individual skeletal maturity indicator but its subjective outcome affects its reliability.
GCF ALP activity can predict accurate pubertal growth period, as it depends up on physiologic bone remodeling; and is efficient and reliable method.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- The diagnostic performance of dental maturity for identification of the circumpubertal growth phases: A meta-analysis. Prog Orthod. 2013;14:8.
- [CrossRef] [PubMed] [Google Scholar]
- The reliability of dental X-ray film in assessment of MP3 stages of the pubertal growth spurt. Am J Orthod Dentofacial Orthop. 1998;114:427-9.
- [CrossRef] [Google Scholar]
- Skeletal maturation evaluation using cervical vertebrae. Am J Orthod Dentofacial Orthop. 1995;107:58-66.
- [CrossRef] [Google Scholar]
- Epiphysial changes in the hand/wrist area as indicators of adolescent stage. Aust Orthod J. 1976;4:87-104.
- [Google Scholar]
- Validity and reliability of a method for assessment of cervical vertebral maturation. Angle Orthod. 2012;82:229-34.
- [CrossRef] [PubMed] [Google Scholar]
- Skeletal Age Assessment Utilizing Cervical Vertebrae. Master's Thesis. University of Pittsburgh; :164.
- [Google Scholar]
- Cervical vertebrae maturation method morphologic criteria: Poor reproducibility. Am J Orthod Dentofacial Orthop. 2011;140:182-8.
- [CrossRef] [PubMed] [Google Scholar]
- Growth hormone (GH) treatment increases serum insulin-like growth factor binding protein-3, bone isoenzyme alkaline phosphatase and forearm bone mineral content in young adults with GH deficiency of childhood onset. Eur J Endocrinol. 1994;131:41-9.
- [CrossRef] [PubMed] [Google Scholar]
- Alkaline phosphatase activity in the plasma of children andadolescents. ClinChem. 1977;23:469-472.
- [CrossRef] [PubMed] [Google Scholar]
- Demonstration of insulin-like growth factor I in human urine. J Clin Endocrinol Metab. 1987;64:1309-12.
- [CrossRef] [PubMed] [Google Scholar]
- The pattern of facial skeletal growth and its relationship to various common indexes of maturation. Am J Orthod Dentofacial Orthop. 2013;143:845-54.
- [CrossRef] [PubMed] [Google Scholar]
- Correlation between salivary and serum markers of bone turnover in osteopenic rats. J Periodontol. 2008;79:158-65.
- [CrossRef] [PubMed] [Google Scholar]
- Differential expression of chemokines and chemokine receptors in inflammatory periapical diseases. Oral Microbiol Immunol. 2005;20:310-6.
- [CrossRef] [PubMed] [Google Scholar]
- Periodontal diagnoses and classification of periodontal diseases. Periodontol 2000. 2004;34:9-21.
- [CrossRef] [PubMed] [Google Scholar]
- Host-derived diagnostic markers for periodontitis: Do they exist in gingival crevice fluid? Periodontol 2000. 2005;39:53-72.
- [CrossRef] [PubMed] [Google Scholar]
- Gingival crevicular fluid alkaline phosphatase activity as a non-invasive biomarker of skeletal maturation. Orthod Craniofac Res. 2011;14:44-50.
- [CrossRef] [PubMed] [Google Scholar]
- Dental maturation is not a reliable indicator of the pubertal growth spurt. Am J Orthod Dentofacial Orthop. 2016;150:4-6.
- [CrossRef] [PubMed] [Google Scholar]
- Development and reliability assessment of a new quality appraisal tool for crosssectional studies using biomarker data (BIOCROSS) BMC Medical Research Methodology. 2018;18:122.
- [CrossRef] [PubMed] [Google Scholar]
- Gingival crevicular fluid alkaline phosphatase activity reflects periodontal healing/recurrent inflammation phases in chronic periodontitis patients. J Periodontol. 2008;79:1200-7.
- [CrossRef] [PubMed] [Google Scholar]
- Assessment of alkaline phosphatase activity in gingival crevicular fluid as a skeletal maturity indicator. Int J Res Med. 2018;7:13-8.
- [Google Scholar]
- Phases of the dentition for the assessment of skeletal maturity: a diagnostic performance study. Am J Orthod Dentofacial Orthop. 2008;133:395-400. quiz 476 e391-2
- [CrossRef] [PubMed] [Google Scholar]
- Dental maturity as an indicator of chronological age: The accuracy and precision of three methods. Eur J Orthod. 1985;7:25-35.
- [CrossRef] [PubMed] [Google Scholar]
- Prospective longitudinal evaluation of the relationship between changes in mandibular length and blood-spot IGF-1 measurements. Am J Orthod Dentofacial Orthop. 2012;141:694-704.
- [CrossRef] [PubMed] [Google Scholar]
- Host enzymes in gingival crevicular fluid as diagnostic indicators of periodontitis. J Clin Periodontol. 1994;21:497-506.
- [CrossRef] [PubMed] [Google Scholar]






