Translate this page into:
Role of articular disc in cartilaginous growth of the mandible in rats
Address for Correspondence: Prof. Kazuo Tanne, Department of Orthodontics and Craniofacial Developmental Biology, Institute of Biomedical and Health Sciences, Hiroshima University, Hiroshima 734-8553, Japan. E-mail: tanne@hiroshima-u.ac.jp
This article was originally published by Wolters Kluwer and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Displacement of the temporomandibular joint (TMJ) disc causes a lateral shift of the mandible and less-developed and/or distally located mandible unilaterally and bilaterally, respectively, if occurred in growing individuals. The purpose of this study was to evaluate mandibular condylar growth in growing rats after TMJ discectomy and to explore a certain significant role of articular disc in the TMJ in mandibular or cartilaginous growth. Eighteen 4-week-old Wistar strain male rats were divided into two groups with nine in each group, i.e., rats with TMJ discectomy (discectomy group) and only sham operation (control group). Four weeks after initiating the experiment, morphometric analyses of the mandible were performed using a rat and mouse cephalometer and micro-computed tomography. Then, the mandibular condyles were subjected to histomorphometric analyses. Condylar and mandibular growth was reduced significantly in the discectomy group than in the control group. In the discectomy group, the condyle also became flatter and smaller. In addition, the 4-layer structure of condylar cartilage was unclear with thicker fibrous and thinner lower hypertrophic layers in the discectomy group when compared to the controls. It is shown that resection of the articular disc substantially affects condylar and mandibular growth in terms of the cartilaginous growth, suggesting that TMJ disc is indispensable for maintaining normal growth of the condyle and mandible, leading to optimal development of the TMJ and the entire mandible.
Keywords
Articular cartilage
cartilaginous growth
mandible
temporomandibular joint disc
temporomandibular joint disorder
INTRODUCTION
The mandibular condyle is an important growth site for the mandible. Condylar growth provides regional adaptive growth with considerable clinical significance, as upward and backward condylar growth displaces the mandible anteriorly and inferiorly.[1]
Mechanical strain on the mandibular condyle controls the size and shape of mandible.[2] In orthodontic treatment, control of condylar growth results in favorable skeletal response and harmonious facial profiles in young patients. On the other hand, in several clinical studies, facial asymmetry and mandibular retrognathia were demonstrated to have an association with coexisting irreducible displacement of temporomandibular joint (TMJ) disc in children and adolescents before and during pubertal growth.[3-7] In animal experiments, TMJ disc displacement was shown to induce histomorphological and structural changes in the articular chondrocytes and condylar cartilage layers and to exert substantial influences on mandibular growth.[8] However, there is little information on the role of TMJ disc in condylar and mandibular growth during the growing period. It is thus hypothesized that a certain role of the TMJ disc in mandibular growth becomes evident when discectomy (complete excision of the articular disc from TMJ) is performed in growing rats. The purpose of this study was to examine the role of TMJ disc in condylar and mandibular growth in growing rats with and without TMJ discectomy and to demonstrate a certain significant role of articular disc in TMJ in cartilaginous growth of the mandible.
MATERIALS AND METHODS
Experimental animals
Eighteen Wistar strain male rats (Charles River Japan, Yokohama, Japan) were divided into two groups with nine in each group, i.e., rats with TMJ discectomy (discectomy group) and only sham operation (control group). At the age of 4 weeks, under general anesthesia with pentobarbital (Somnopentyl; Kyoritsu, Tokyo, Japan), bilateral discectomy was performed in the discectomy group.
All rats were fed a soft diet (CE-2; Japan Clea Co., Tokyo, Japan) with water ad libitum. Body weight was measured for every 2–3 days during the experiment and it was used as an indicator of general growth. Rats were sacrificed by perfusion fixation under general anesthesia with pentobarbital (Somnopentyl; Kyoritsu) 4 weeks after surgery. All rats used in this study were maintained at the Natural Science Center, Hiroshima University, and handled in conformity with the ARRIVE guidelines for animal research[9] and the Rules for Animal Experiments in Hiroshima University.
Bilateral discectomy
TMJ disc excision was performed under general anesthesia with pentobarbital (Somnopentyl; Kyoritsu). A horizontal incision was made 5–7 mm below the zygomatic arch, exposing the superficial masseter muscle fibers. These were then separated by blunt and sharp dissection until mandibular ramus and condyle were exposed.
TMJ discs were fully exposed and carefully resected with scissors and a knife without producing any injury to the articular surface. Close inspection ensured a clean and complete discectomy on both sides. Muscle fibers were returned to the original position and the incision was sutured firmly. These procedures were performed under sterile conditions.
Morphometric analysis of the mandible by the use of lateral cephalograms
Four weeks after initiating the experiment, lateral cephalograms were taken of all rats using a rat and mouse cephalometer (Asahi Roentgen, Kyoto, Japan). The head of each animal was fixed and halved on the median sagittal plane. The halved head was placed on dental occlusal film (DF-50; Eastman Kodak, Rochester, NY, USA), and lateral cephalograms were taken under electronic controls of 6 mA and 30–35 Kvp with an exposure time of 3.0 s.
On the lateral cephalograms, landmarks and measurement items were established according to the method of Fujita et al.[10] [Figure 1a]. Morphometric analysis of the mandible was performed three times for each of the measurement items.
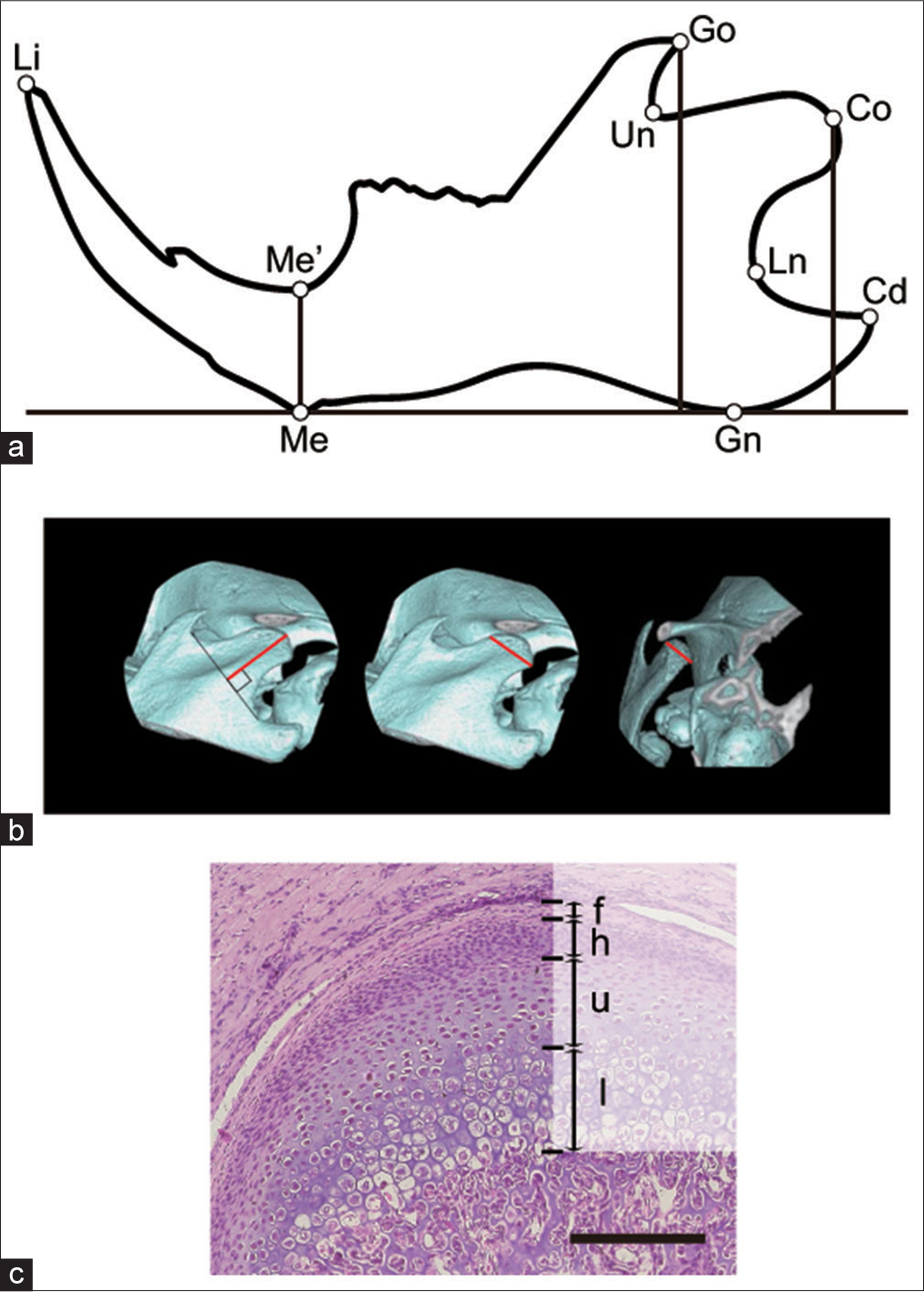
- (a) Landmarks and measurement items used for cephalometric analyses. Landmarks: Me: Point on the most inferior contour of the lower border of mandible, adjacent to incisors, Go: Most posterior point of angular process of mandible, Cd: Most posterosuperior point of condylar process, Co: Most posterior point of coronoid process of mandible, Un: Point in the deepest part of the upper mandibular notch curvature, Ln: Point in the deepest part of the lower mandibular notch curvature, Me’: Point in the deepest part of lingual alveolar bone, Gn: Point on the most inferior contour of angular process of mandible, Li: Most prominent point on the incisal edge of the lower incisor. Measurement items: Me-Go, Me-Cd, Me-Co, Me-Un, Me-Ln, Me-Gn, and Me-Li: Size of mandible. MeH, CoH, and CdH: Vertical size of mandible measured on the Me-Gn baseline to Me’, Co, and Cd. (b) Micro-computed tomography images of the rat condyle. Condylar length (left), width (center), and thickness (right) measured on each micro-computed tomography image. (c) H and E stained section of rat temporomandibular joint. Cartilage layer was thickened in the center of the condyle (f = fibrous cell layer, h = proliferating cell layer, u = upper hypertrophic layer, l = lower hypertrophic layer). Bar = 100 μm
Morphometric analysis of the condyles by the use of micro-computed tomography
We used micro-computed tomography (CT) (R_mCT®; Rigaku Co., Tokyo, Japan) at a magnification of 6.7 fold (voxel size: 50 μm3), tube voltage of 90 kv, and tube current of 50 μA. The image reconstruction software used was i-view-R® (J. Morita Mfg. Corp., Kyoto, Japan). The field of view was 24 mm × 24 mm × 24 mm (pixel number: 480 × 480 × 480), and scanning time was 17 s. Micro-CT examination was performed for the condyle at 4 weeks after initiating the experiment.
Micro-CT data of the condyle were analyzed with i-view-R® (J. Morita Mfg. Corp.). The TMJ was sectioned along the frontal, axial, and sagittal planes for longitudinal comparison of the three-dimensional architecture, following the method of Kuroki et al.[11] The frontal plane was determined by searching for the thickest region of the condylar head relative to the reconstructed three-dimensional condylar images. The sagittal plane was adjusted on frontal sections in order that the plane passes perpendicularly through the center of the condyle. The axial plane was adjusted similarly on the frontal and sagittal sections in order that the planes pass perpendicularly through the center of the condylar head. Through these processes, the best-matched frontal, sagittal, and axial images were determined at each time point during measurements.
The length and mesiolateral and rostrocaudal widths of the condyle were measured on the condylar images [Figure 1b]. Changes in the size of the condyle were determined by calculating the absolute difference between actual measurement values on the TMJ images obtained before and after discectomy.
Histomorphometric analysis
Four weeks after the beginning of the experiment, both TMJs were dissected out and placed in formalin. Specimens were decalcified in ethylenediaminetetracetic acid, cut into sagittal sections of 7 μm thickness, and finally stained with hematoxylin and eosin (H and E) for histological evaluation.
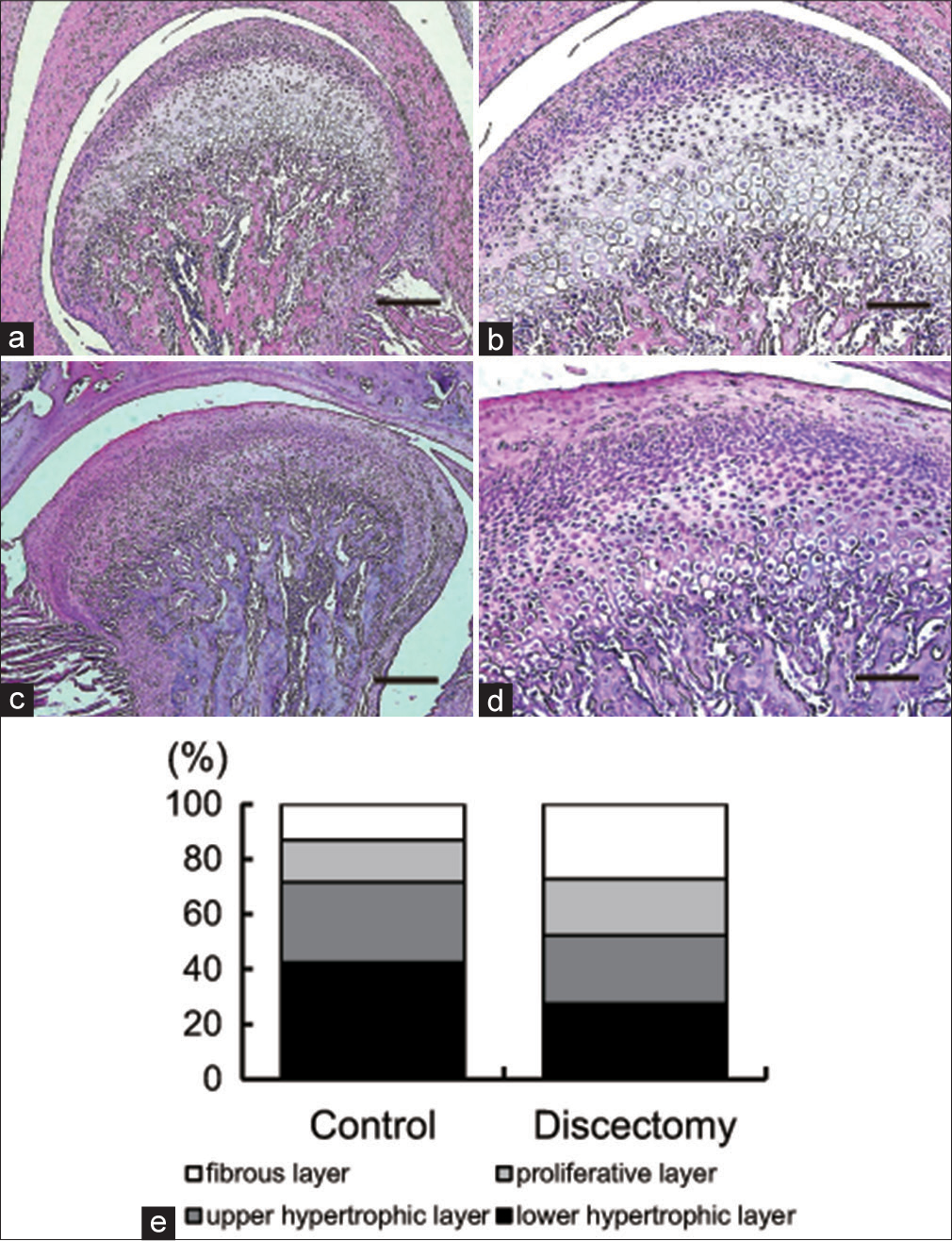
From histologic images, four cartilage layers were distinguished, i.e., fibrous, proliferative, upper hypertrophic, and lower hypertrophic layers, based on the method described by Xiong et al.[12] [Figure 1c]. The thickness of each layer was quantified using a computer-assisted image analyzing system with a digital camera (Biozero, Keyence, Osaka, Japan) and analysis software (BZ-II Analyzer, Keyence). The thickness of the cartilage layers in condyles was examined by digital image analysis and compared between the discectomy and control groups.
Statistical analysis
Unpaired t-test was used to examine the mean differences between the two groups at a confidence level of >95%.
RESULTS
Changes in body weight throughout the experiment
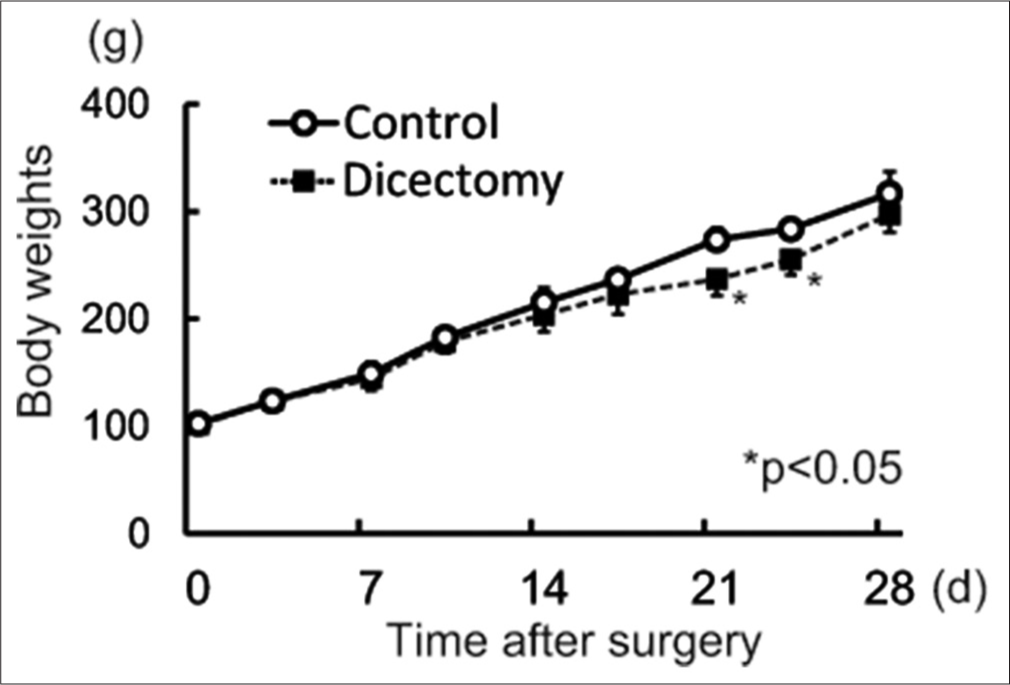
In the discectomy group, body weight increased from 101 to 297 g during the experiment, whereas the value changed from 103 to 317 g in the control group. Although there was a significant difference in body weight between the groups at 21 and 24 days after surgery, no significant differences were observed at the remaining time points including the end of experiment, indicating no substantial influences of a series of experiment on the entire general growth of rats with TMJ discectomy [Appendix 1].
Morphometric findings from cephalometric analyses
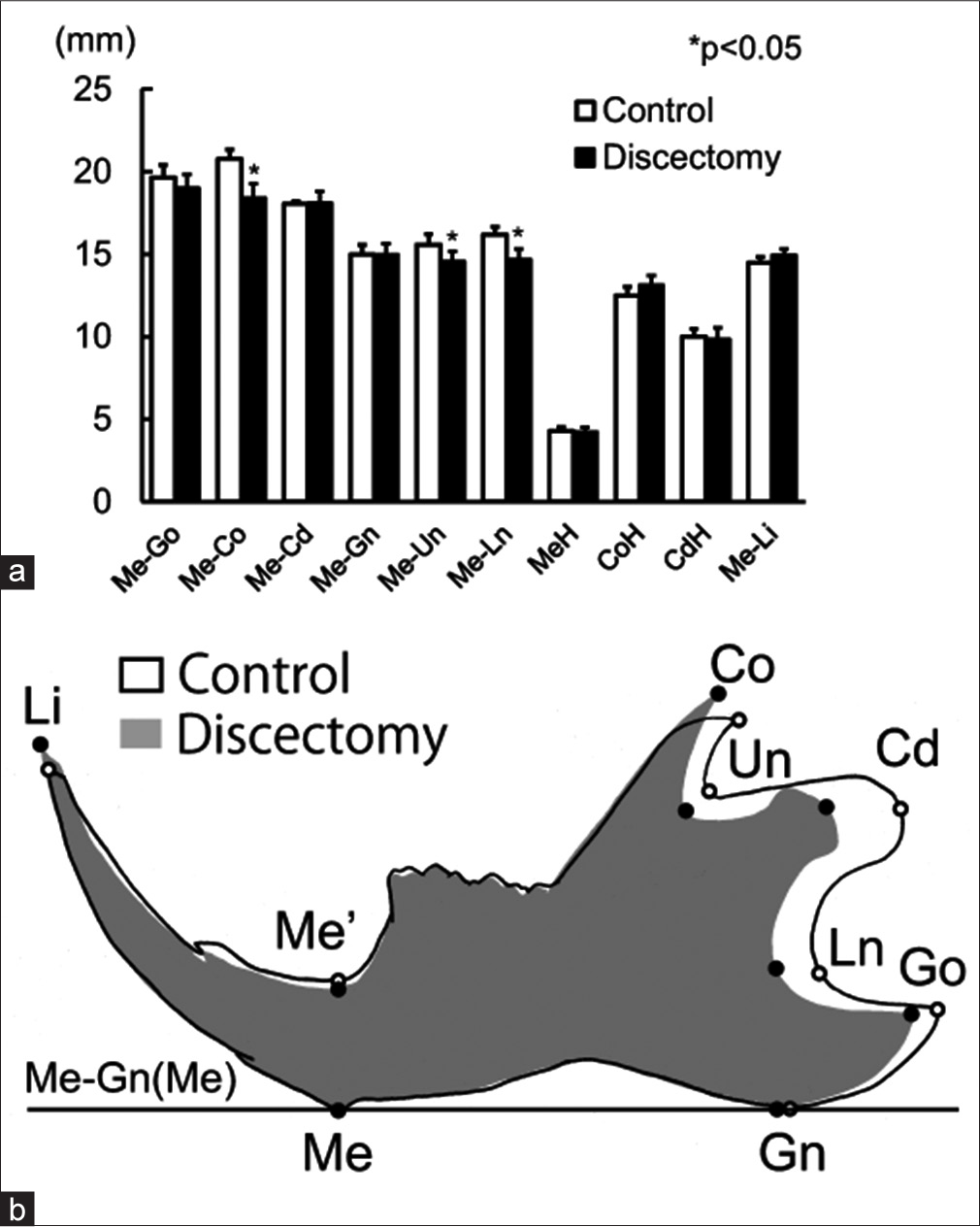
On the lateral cephalograms, in the discectomy group, Me-Co, Me-Un, and Me-Ln were significantly smaller than in the controls. On the other hand, Me-Li was significantly greater in the discectomy group than in the control group [Figure 2a]. No significant differences in the size of the mandible were observed between the discectomy and control groups; however, morphologic differences were observed prominently on superimposed lateral cephalogram tracings between the discectomy and control groups [Figure 2b].
- (a) Comparison of measured values between discectomy and control groups. (b) Superimposition of the mean tracings on the Me-Gn line at Me. Control group (white) versus discectomy group (gray)
Morphometric findings from micro-computed tomography analyses
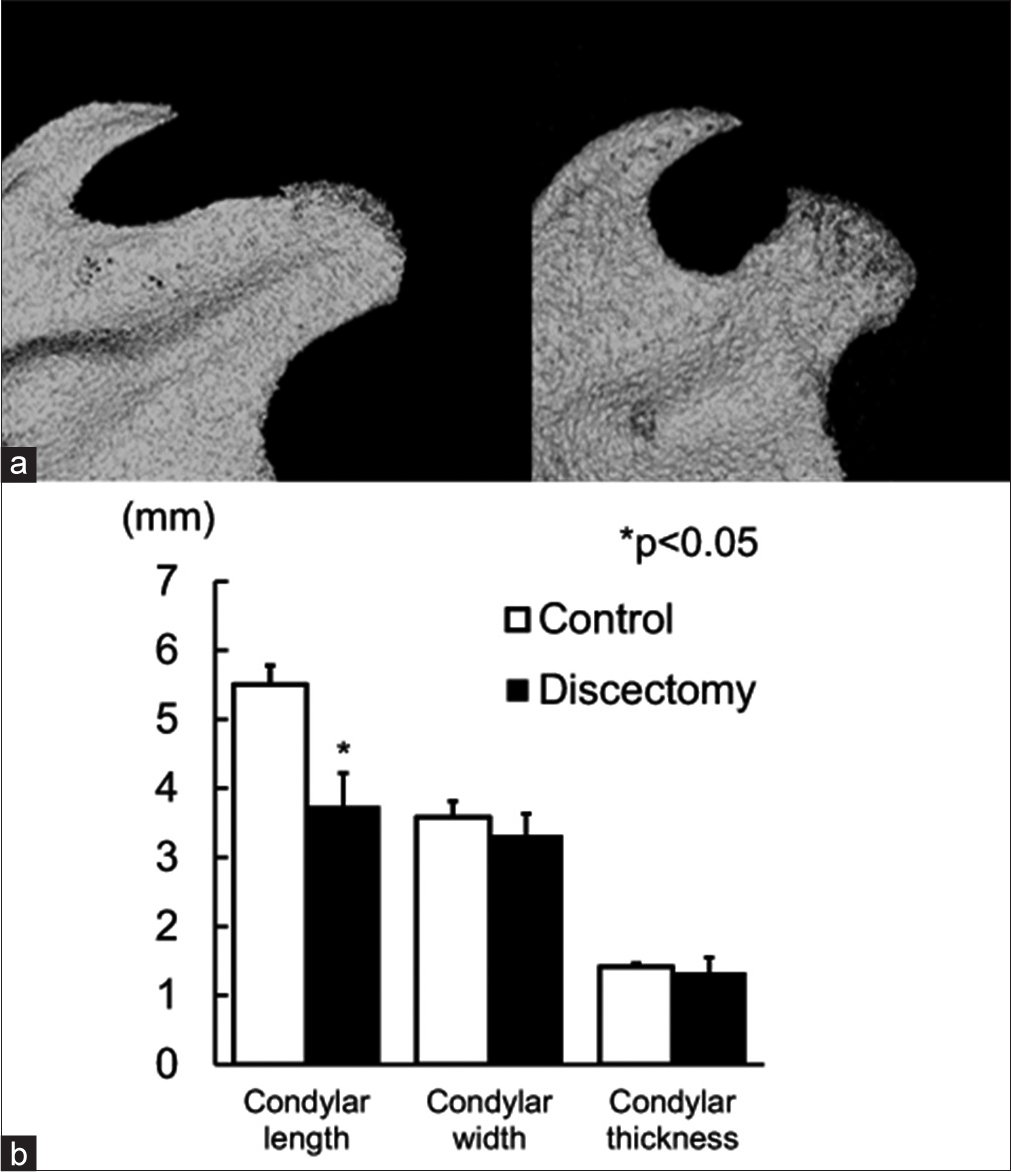
Condyles of the experimental group exhibited morphologic changes on day 28 on micro-CT image. The most remarkable difference was observed on the shape of condyle; the condyles were flatter in the discectomy group than in the controls [Figure 3a]. A significant difference in condylar length was also observed between the two groups on day 28 [Figure 3b].
- (a) Micro-computed tomography image of control group (left side) and discectomy group (right side). (b) Comparison of measured values on micro-computed tomography images between discectomy and control groups
Histomorphometric findings
Figure 4 shows H and E stained sagittal TMJ sections in the discectomy and control rats. In controls, the condyles exhibited a round and smooth articular surface without any pathologic sign. Four different layers of the cartilage were clearly distinguished (fibrous, proliferative, upper hypertrophic. and lower hypertrophic layers), and cells were regularly arranged.
- H and E stained sections of rat condyle. (a) Control condyle. (b) Higher magnification of (a). (c) Discectomy group temporomandibular joint at 4 weeks after surgery. The shape of condyle was flatter than the control condyle. (d) Higher magnification of (c). Lower hypertrophic layer was thinner when compared to the control group. (e) Condylar cartilage layer. (a and c), bar = 300 μm; (b and d) bar = 100 μm
On the other hand, the condyle became flatter in the discectomy group than in the controls. The surface of condyle was rougher in the discectomy group than in the controls. The thickness of condylar cartilage layers also became smaller when compared to the normal condyles. Although four layers of the condylar cartilage were also detected in discectomized condyles, the boundary zone between layers was unclear. The fibrous and lower hypertrophic layers were significantly thicker and thinner than in the controls, respectively [Figure 4e].
DISCUSSION
As soft diet induces less influence on the response of condylar cartilage than hard diet in rats with TMJ discectomy,[13] in this study, rats were fed a soft diet. Male rats aged 4 weeks were used because their growth rate is markedly high.[14] Four-week observation period defined in this study is considered appropriate because mandibular retrognathia generally appeared approximately 30 days after the initiation of experiment in a previous study.[15] Although a certain degree of weight loss was observed in all rats with bilateral TMJ discectomy during the later stage of experiment, no significant differences were found between the discectomy and control groups at the end of experiment, indicating that the experimental procedures exerted no substantial influences on the general growth of rats.
In this study, TMJ discectomy resulted in condylar deformity such as flattening and thicker fibrous layer of the condylar cartilage.[8] The TMJ disc is <1 mm thick in rats. However, the disc plays various important roles in reducing friction between condyle and glenoid fossa and resisting TMJ loading of 10 MPa at maximum.[16-18] It is well known that the load distribution capacity is dependent on disc thickness.[19] Movement over the surface of the articular disc produces mechanical forces.[20] In fact, excessive mouth opening induces TMJ remodeling in rats.[21,22] From the present histomorphometric analyses, a deformed condyle was observed more prominently and frequently in the discectomy group than in controls. The reason may be because nonphysiological compression stress or strain was loaded onto the condyle without the TMJ disc during condylar movement for mastication. Previous studies using growing animals also demonstrated morphological changes such as chondrocyte proliferation and enlargement of articular cartilage layers.[23-25] In addition, such compression stress or strain reduced cartilaginous growth of the mandibular condyle.[26] From these findings, it would be a reasonable assumption that physiological force during jaw movement is an important factor for the optimal development of cartilage in the TMJ and the disc may have an essential ability to partially control mechanical stresses on the condyle during the growing period.
Since condylar cartilage is sensitive to external mechanical stimuli,[27] these results are assumed to be dependent on changes in the mechanical loading acting on the condyle. The present study has suggested degenerative changes in condylar cartilage occurred by a certain nonphysiological mechanical loading following TMJ discectomy in growing rats. Recent animal studies have also demonstrated that nonuniform and reduced mandibular growth was highly associated with histological changes in the condylar cartilage.[8] It is thus assumed that after TMJ discectomy the joint space surely becomes smaller and two bony components are compressed more substantially at the articulating surfaces. Compression of synovial cells was shown to enhance osteoclast formation, which further induces osteoclastic bone resorption in the TMJ.[28] In animal experiments, extensive peripheral osteoclast activity without inflammation was observed in the mandibular condyle with discectomy.[29] These findings suggest that nonuniform and excessive mechanical loading induced by discectomy in the TMJ negatively affects condylar and mandibular growth, if induced before and during pubertal growth. From these considerations, it is demonstrated that maintaining the proper position of TMJ disc on the condyle by a certain conservative therapy with the use of occlusal splint is of a significant importance for optimal growth of the condyle and entire mandible for growing patients with internal derangement of TMJ.[30]
CONCLUSIONS
Bilateral TMJ discectomy in growing rats causes significant reductions in mandibular growth
Defects in the articular disc influence mandibular growth, suggesting that the articular disc is crucial in condylar and mandibular growth
Intra-articular pathology associated with disc displacement may exert adverse effects on growth and development of the craniofacial skeleton, the mandible in particular, and the subsequent outcome of orthodontic treatment aiming appropriate skeletal growth control.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Growth and remodeling of the human maxilla. Am J Orthod. 1965;51:446-64.
- [CrossRef] [Google Scholar]
- Runx2 regulates endochondral ossification in condyle during mandibular advancement. J Dent Res. 2005;84:166-71.
- [CrossRef] [PubMed] [Google Scholar]
- Comparison of skeletal and dental morphology in asymptomatic volunteers and symptomatic patients with bilateral disk displacement without reduction. Angle Orthod. 2004;74:684-90.
- [Google Scholar]
- Relationship between functional disc position and mandibular displacement in adolescent females: Posteroanterior cephalograms and magnetic resonance imaging retrospective study. J Oral Rehabil. 2002;29:417-22.
- [CrossRef] [PubMed] [Google Scholar]
- Adolescent female craniofacial morphology associated with advanced bilateral TMJ disc displacement. Eur J Orthod. 1998;20:701-12.
- [CrossRef] [PubMed] [Google Scholar]
- Pediatric internal derangements of the temporomandibular joint: Effect on facial development. Am J Orthod Dentofacial Orthop. 1993;104:51-9.
- [Google Scholar]
- Condylar bony change and craniofacial morphology in orthodontic patients with temporomandibular disorders (TMD) symptoms: A pilot study using helical computed tomography and magnetic resonance imaging. Clin Orthod Res. 1999;2:133-42.
- [Google Scholar]
- Cartilage changes link retrognathic mandibular growth to TMJ disc displacement in a rabbit model. Int J Oral Maxillofac Surg. 2011;40:621-7.
- [Google Scholar]
- Improving bioscience research reporting: The ARRIVE guidelines for reporting animal research. Osteoarthritis Cartilage. 2012;20:256-60.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of sex hormone disturbances on craniofacial growth in newborn mice. J Dent Res. 2004;83:250-4.
- [CrossRef] [PubMed] [Google Scholar]
- In vivo morphometric analysis of inflammatory condylar changes in rat temporomandibular joint. Oral Dis. 2011;17:499-507.
- [CrossRef] [PubMed] [Google Scholar]
- Mechanical strain leads to condylar growth in adult rats. Front Biosci. 2005;10:67-73.
- [Google Scholar]
- Effect of postoperative diet on condylar cartilage response to discectomy. J Oral Maxillofac Surg. 1997;55:1259-64.
- [Google Scholar]
- The growth of the muscles of mastication in the rat. J Anat. 1972;113:391-408. Pt 3
- [CrossRef] [Google Scholar]
- Effects of altered mandibular function on mandibular growth after condylectomy. Eur J Orthod. 1997;19:9-19.
- [CrossRef] [PubMed] [Google Scholar]
- Biomechanical properties and collagen fiber orientation of temporomandibular joint discs in dogs: 2. Tensile mechanical properties of the discs. J Craniomandib Disord. 1991;5:107-14.
- [Google Scholar]
- Strain rate dependent orthotropic properties of pristine and impulsively loaded porcine temporomandibular joint disk. J Biomed Mater Res. 2001;57:25-34.
- [CrossRef] [Google Scholar]
- Biomechanical behavior of the temporomandibular joint disc. Crit Rev Oral Biol Med. 2003;14:138-50.
- [CrossRef] [Google Scholar]
- In vitro measurement of the stress-distribution properties of the pig temporomandibular joint disc. Arch Oral Biol. 1994;39:439-48.
- [CrossRef] [Google Scholar]
- Stress-field translation in the healthy human temporomandibular joint. J Dent Res. 2000;79:1740-6.
- [CrossRef] [PubMed] [Google Scholar]
- Vascular endothelial growth factor plays an important autocrine/ paracrine role in the progression of osteoarthritis. Histochem Cell Biol. 2005;123:275-81.
- [CrossRef] [Google Scholar]
- Jaw-muscle activity changes after the induction of osteoarthrosis in the temporomandibular joint by mechanical loading. J Orofac Pain. 2008;22:153-62.
- [CrossRef] [Google Scholar]
- Effect of mechanical forces on chondrocyte maturation and differentiation in the mandibular condyle of the rat. J Dent Res. 1994;73:1150-6.
- [CrossRef] [PubMed] [Google Scholar]
- Growth and type-II collagen expression in the glenoid fossa of the temporomandibular joint during altered loading: A study in the rat. Eur J Orthod. 1996;18:3-9.
- [Google Scholar]
- Chondrocyte proliferation of the cranial base cartilage upon in vivo mechanical stresses. J Dent Res. 2002;81:701-5.
- [CrossRef] [PubMed] [Google Scholar]
- The adaptive remodeling of condylar cartilage – A transition from chondrogenesis to osteogenesis. J Dent Res. 2005;84:691-9.
- [Google Scholar]
- Effects of a mandibular lateral shift on the condyle and mandibular bone in growing rats. Angle Orthod. 2007;77:787-93.
- [Google Scholar]
- Compressive mechanical stress promotes osteoclast formation through RANKL expression on synovial cells. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103:334-41.
- [Google Scholar]
- The effects of unilateral discectomy and condylectomy on the contralateral intact rabbit craniomandibular joint. J Oral Maxillofac Surg. 2006;64:1261-6.
- [CrossRef] [PubMed] [Google Scholar]
- Current status of temporomandibular joint disorders and the therapeutic system derived from a series of biomechanical, histological and biochemical studies. APOS Trends Orthod. 2015;5:4-21.
- [Google Scholar]
APPENDIX
- Changes in body weight in the discectomy and control groups






